Author Interviews, Flu - Influenza, Pediatrics, Pediatrics, Vaccine Studies / 15.06.2020
Parents Remain Concerned About Childhood Vaccines, Especially for Flu
MedicalResearch.com Interview with:
[caption id="attachment_54587" align="alignleft" width="175"] Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination
Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination
 Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination
Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination 

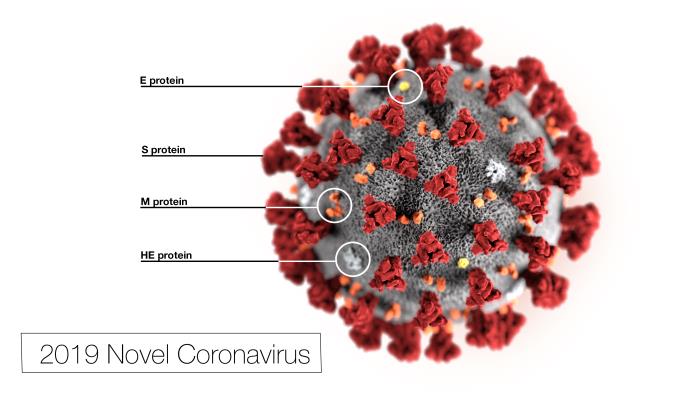
 Dr. Emily Parker Hyle[/caption]
Emily Parker Hyle, M.D.
Assistant Professor of Medicine
Massachusetts General Hospital
MedicalResearch.com: What is the background for this study?
Response: We found that many children who were planning to travel internationally were eligible for MMR vaccination prior to departure but often did not receive it - especially if they were aged 6 months to 6 years. That is because most children do not routinely receive their first dose of MMR till 12-15 months of age and their second dose of MMR till 4-6 years of age. However, ACIP recommendations are different for children who are traveling internationally. The risk of being infected with measles is much higher outside of the US, so it is recommended that children older than 1 year have had 2 MMR vaccinations and that children 6-12 months receive 1 MMR vaccination prior to travel. MMR vaccination is a safe and effective way to greatly reduce the risk of measles infection.
Dr. Emily Parker Hyle[/caption]
Emily Parker Hyle, M.D.
Assistant Professor of Medicine
Massachusetts General Hospital
MedicalResearch.com: What is the background for this study?
Response: We found that many children who were planning to travel internationally were eligible for MMR vaccination prior to departure but often did not receive it - especially if they were aged 6 months to 6 years. That is because most children do not routinely receive their first dose of MMR till 12-15 months of age and their second dose of MMR till 4-6 years of age. However, ACIP recommendations are different for children who are traveling internationally. The risk of being infected with measles is much higher outside of the US, so it is recommended that children older than 1 year have had 2 MMR vaccinations and that children 6-12 months receive 1 MMR vaccination prior to travel. MMR vaccination is a safe and effective way to greatly reduce the risk of measles infection. 



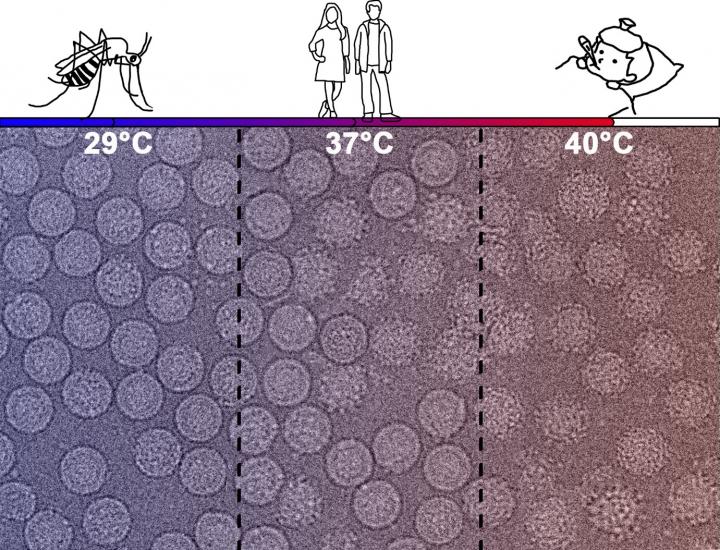
 Prof. Satchi-Fainaro[/caption]
Prof. Ronit Satchi-Fainaro, PhD
Head, Cancer Research and Nanomedicine Laboratory
Department of Physiology and Pharmacology,
Sackler Faculty of Medicine,
Tel Aviv University,
Tel-Aviv 69978, Israel
[caption id="attachment_50670" align="alignleft" width="116"]
Prof. Satchi-Fainaro[/caption]
Prof. Ronit Satchi-Fainaro, PhD
Head, Cancer Research and Nanomedicine Laboratory
Department of Physiology and Pharmacology,
Sackler Faculty of Medicine,
Tel Aviv University,
Tel-Aviv 69978, Israel
[caption id="attachment_50670" align="alignleft" width="116"] Prof. Florindo[/caption]
Prof. Helena Florindo, PhD
Head, BioNanoSciences – iMed.ULisboa
Faculty of Pharmacy,
University of Lisbon
Lisbon, Portugal
MedicalResearch.com: What are the main findings?
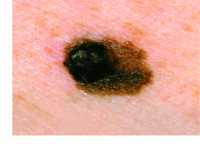
Response: The war against cancer in general, and melanoma in particular, has advanced over the years through a variety of treatment modalities, such as surgery, chemotherapy, radiation therapy and immunotherapy. The immune checkpoint inhibitors brought a breakthrough solution for advanced melanoma patients, but only a low percentage of those respond to this therapy, developing resistance and being affected by severe side effects. Despite the success of several vaccines against viral diseases, this success has not been materialized yet against cancer.
This study led by my lab at Tel Aviv University, and Helena Florindo’s lab at the University of Lisbon, describes the development of an effective nano-vaccine against melanoma, that also sensitizes the immune system to immunotherapies.
This nano-vaccine prevented melanoma, and also led to remarkable tumor inhibition and prolonged survival in mice already affected by this disease.
Prof. Florindo[/caption]
Prof. Helena Florindo, PhD
Head, BioNanoSciences – iMed.ULisboa
Faculty of Pharmacy,
University of Lisbon
Lisbon, Portugal
MedicalResearch.com: What are the main findings?
Response: The war against cancer in general, and melanoma in particular, has advanced over the years through a variety of treatment modalities, such as surgery, chemotherapy, radiation therapy and immunotherapy. The immune checkpoint inhibitors brought a breakthrough solution for advanced melanoma patients, but only a low percentage of those respond to this therapy, developing resistance and being affected by severe side effects. Despite the success of several vaccines against viral diseases, this success has not been materialized yet against cancer.
This study led by my lab at Tel Aviv University, and Helena Florindo’s lab at the University of Lisbon, describes the development of an effective nano-vaccine against melanoma, that also sensitizes the immune system to immunotherapies.
This nano-vaccine prevented melanoma, and also led to remarkable tumor inhibition and prolonged survival in mice already affected by this disease. 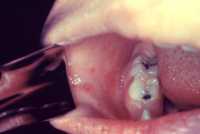
 Dr. Nandi[/caption]
Arindam Nandi PhD
Center for Disease Dynamics, Economics & Policy
MedicalResearch.com: What is the background for this study?
Response: The motivation for this study comes from a small but growing body of evidence on the potential long-term benefits of vaccines. The recent resurgence of measles outbreaks in several countries which had previously eliminated the virus makes our study additionally relevant. There have been over 1,000 measles cases reported across 28 states in the US so far in 2019, which is the largest number of cases the country has seen in almost 3 decades. Similarly high number of cases have been reported in several European countries in recent years. This study reiterates the importance of vaccination and proves the long-term benefits of the measles vaccine in low- and middle-income countries, which account for a large proportion of global measles cases.
Dr. Nandi[/caption]
Arindam Nandi PhD
Center for Disease Dynamics, Economics & Policy
MedicalResearch.com: What is the background for this study?
Response: The motivation for this study comes from a small but growing body of evidence on the potential long-term benefits of vaccines. The recent resurgence of measles outbreaks in several countries which had previously eliminated the virus makes our study additionally relevant. There have been over 1,000 measles cases reported across 28 states in the US so far in 2019, which is the largest number of cases the country has seen in almost 3 decades. Similarly high number of cases have been reported in several European countries in recent years. This study reiterates the importance of vaccination and proves the long-term benefits of the measles vaccine in low- and middle-income countries, which account for a large proportion of global measles cases.