ASCO, AstraZeneca, Author Interviews, Cancer Research, Melanoma / 13.06.2020
ASCO20: IMFINZI plus Chemotherapy For Extensive-Stage Small Cell Lung Cancer
MedicalResearch.com Interview with:
[caption id="attachment_54541" align="alignleft" width="200"] Dr. Chen[/caption]
Yuanbin Chen, MD, PhD
Cancer & Hematology
Centers of Western Michigan
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Chen[/caption]
Yuanbin Chen, MD, PhD
Cancer & Hematology
Centers of Western Michigan
MedicalResearch.com: What is the background for this study? What are the main findings?
 Dr. Chen[/caption]
Yuanbin Chen, MD, PhD
Cancer & Hematology
Centers of Western Michigan
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Chen[/caption]
Yuanbin Chen, MD, PhD
Cancer & Hematology
Centers of Western Michigan
MedicalResearch.com: What is the background for this study? What are the main findings?
-
- Response: The CASPIAN trial was a randomized, open-label, multi-center global Phase III trial in the first-line treatment of patients with extensive-stage small cell lung cancer (ES-SCLC). The trial compared IMFINZI in combination with etoposide and either carboplatin or cisplatin chemotherapy, or IMFINZI and chemotherapy with the addition of a second immunotherapy, tremelimumab, versus chemotherapy alone – the primary endpoint being overall survival (OS). After a median follow up of more than two years, the latest results for IMFINZI plus chemotherapy demonstrate a sustained and clinically meaningful OS benefit for patients with extensive-stage small cell lung cancer (ES-SCLC), maintaining a 25% reduction in the risk of death versus chemotherapy alone. Updated median OS was 12.9 months versus 10.5 for chemotherapy.
- In a post-hoc analysis, 22.2% of patients treated with IMFINZI plus chemotherapy remained alive after 24 months, versus 14.4%, for chemotherapy alone.
- Post-hoc analysis also showed that for IMFINZI plus chemotherapy, 11.0% of patients were alive and progression-free at 24 months versus 2.9% for chemotherapy alone.
- IMFINZI plus chemotherapy maintained a high confirmed objective response rate (ORR) (68% versus 58%) and in a post-hoc analysis, duration of response (DoR) for IMFINZI at 24 months was 13.5% versus 3.9% for chemotherapy alone.
- At 24 months, 12% of patients in the IMFINZI plus chemotherapy arm remained on IMFINZI treatment.]
- Response: The CASPIAN trial was a randomized, open-label, multi-center global Phase III trial in the first-line treatment of patients with extensive-stage small cell lung cancer (ES-SCLC). The trial compared IMFINZI in combination with etoposide and either carboplatin or cisplatin chemotherapy, or IMFINZI and chemotherapy with the addition of a second immunotherapy, tremelimumab, versus chemotherapy alone – the primary endpoint being overall survival (OS). After a median follow up of more than two years, the latest results for IMFINZI plus chemotherapy demonstrate a sustained and clinically meaningful OS benefit for patients with extensive-stage small cell lung cancer (ES-SCLC), maintaining a 25% reduction in the risk of death versus chemotherapy alone. Updated median OS was 12.9 months versus 10.5 for chemotherapy.





 Dr. Garstang[/caption]
Dr Joanna Garstang
Consultant Community Paediatrician / Designated Doctor for Child Death
Allenscroft Children's Centre
Kings Heath, Birmingham UK
MedicalResearch.com: What is the background for this study?
Response: Each year in England and Wales there around 3-400 babies die unexpectedly, in many cases the cause of death remains unexplained and these deaths are called Sudden Infant Death Syndrome (SIDS). Parents are understandably anxious about the risks for future children, the Care of Next Infant (CONI) programme was set up to offer support for families. In this study, we looked at records of families registered with CONI between 2000-2015 to determine the frequency and causes for repeat unexpected infant deaths.
Dr. Garstang[/caption]
Dr Joanna Garstang
Consultant Community Paediatrician / Designated Doctor for Child Death
Allenscroft Children's Centre
Kings Heath, Birmingham UK
MedicalResearch.com: What is the background for this study?
Response: Each year in England and Wales there around 3-400 babies die unexpectedly, in many cases the cause of death remains unexplained and these deaths are called Sudden Infant Death Syndrome (SIDS). Parents are understandably anxious about the risks for future children, the Care of Next Infant (CONI) programme was set up to offer support for families. In this study, we looked at records of families registered with CONI between 2000-2015 to determine the frequency and causes for repeat unexpected infant deaths.
![MedicalResearch.com Interview with: Sarah Messiah, PhD, MPH Professor of epidemiology, human genetics, and environmental sciences UTHealth School of Public Health Dallas MedicalResearch.com: What is the background for this study? Response: Given that obesity is a prevalent, serious, complex, chronic, and relapsing disease, and severe obesity is a deepening crisis, it is important to pay special attention to these challenges during the COVID-19 pandemic. This will avoid placing an even greater burden on individuals, health systems, and society in the post-COVID-19 recovery phase. Thus, it is critically important to document, in real time, how socioecological determinants of health are impacting behaviors among those with obesity. Before our study, how the COVID-19 pandemic is impacting weight management, health behaviors, and psychosocial health in particular among people with obesity was unknown. MedicalResearch.com: What are the main findings? Response: A total of 123 patients (87% female, mean age 51.2 years, mean BMI 40.2, 49.2% Non-Hispanic white, 28.7% Non-Hispanic black, 16.4% Hispanic, 7% other ethnicity, 33.1% completed bariatric surgery were included). -Two patients tested positive for SARS-CoV-2 and 14.6% reported symptoms. -10% lost their job since the beginning of the pandemic -25% reported food insecurity in that they sometimes, or always could not afford to eat balanced meals -72.8% reported increased anxiety and 83.6% increased depression since stay-at-home orders were initiated. -69.6% reported more difficultly in achieving weight loss goals -47.9% reported less exercise time and 55.8% reported less intensity -49.6% reported increased stockpiling of food -61.2% reported stress eating -61.2% reported following healthy diet plans more challenging - Hispanics were less likely to report anxiety versus non-Hispanic whites MedicalResearch.com: What should readers take away from your report? Response: In a relatively short period of time, our results show that there have been substantial perceptions in changes in health behaviors among patients with obesity. Our sample described a decreases in positive health behaviors, increases in deleterious behaviors and associated deterioration in mental health. Even though actual COVID-19 disease burden was low (1.7% tested positive for COVID-19 and another 14.6% reported symptoms), the pandemic is having a significant impact on those without infections. The major strength of this study is that it is the first snapshot into how the COVID-19 pandemic has influenced health behaviors for patients with obesity. Prior to the pandemic, it has been reported that Americans consume 20% of their calories from restaurants and that there are worsening disparities in fast food meal quality by race/ethnicity, education, and income. Due to recent economic challenges, patients may be more likely to select cheaper foods, which are often energy dense and nutrient poor. Therefore, even though we found more patients are cooking at home, the type of foods that are being stockpiled are likely to be processed foods due to their longer shelf life. These are associated with greater intake of fat, carbohydrate, and calories, which facilitate greater weight gain when compared to more balanced diets. It is possible Americans may be trading one pattern of low-quality consumption for another by choosing cheaper processed foods instead of eating out. MedicalResearch.com: What recommendations do you have for future research as a result of this work? Response: These results have implications for clinical practice and management of patients with obesity as we now move into post-COVID-19 relief efforts. Due to the increase in obesogenic behaviors related to the COVID-19 pandemic that were found here, it is paramount that healthcare access is not disrupted for patients with obesity. Maintaining these vital services will prevent exacerbating the negative health and economic consequences of excess body weight. This includes access to primary care providers, Obesity Medicine specialists and bariatric surgery programs. In addition to asking about diet and exercise patterns, screening for indicators of mental health, loneliness, financial stressors and behaviors that may influence body weight should be implemented by healthcare teams to combat this problem. Disclosures: This work was funded by the National Institutes of Health, National Institute on Minority Health and Health Disparities (Grant #R01MD011686). This work was a joint collaboration among investigators from UTHealth School of Public Health, UT Southwestern Medical Center and Minimally Invasive Surgical Associates. Citation: Almandoz, J.P., Xie, L., Schellinger, J.N., Mathew, M.S., Gazda, C., Ofori, A., Kukreja, S. and Messiah, S.E. (2020), Impact of COVID ‐19 Stay‐at‐Home Orders on Weight‐Related Behaviors Among Patients with Obesity. Clin Obes. Accepted Author Manuscript. doi:10.1111/cob.12386 [subscribe] [last-modified] The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2020/06/Dr-Sarah-E.-Messiah.jpg)





 Dr. Travers[/caption]
Colm Travers, M.D., MSPH
Assistant Professor
Department of Pediatrics
University of Alabama at Birmingham
MedicalResearch.com: What is the background for this study?
Response: It is known that black mothers are much more likely to deliver preterm and low birth weight infants. The purpose of this study was to determine whether racial/ethnic disparities in care practices and outcomes were decreasing or increasing among extremely preterm infants.
These are infants born from 22 to 27 weeks of gestation who have a high risk of death and major illnesses.
Dr. Travers[/caption]
Colm Travers, M.D., MSPH
Assistant Professor
Department of Pediatrics
University of Alabama at Birmingham
MedicalResearch.com: What is the background for this study?
Response: It is known that black mothers are much more likely to deliver preterm and low birth weight infants. The purpose of this study was to determine whether racial/ethnic disparities in care practices and outcomes were decreasing or increasing among extremely preterm infants.
These are infants born from 22 to 27 weeks of gestation who have a high risk of death and major illnesses. 


 Ning Ding[/caption]
Ning Ding MPH, PhD candidate
[caption id="attachment_54465" align="alignleft" width="141"]
Ning Ding[/caption]
Ning Ding MPH, PhD candidate
[caption id="attachment_54465" align="alignleft" width="141"]








 Dr. Kao-Ping Chua[/caption]
Kao-Ping Chua, M.D., Ph.D.
Assistant Professor, Pediatrics, Medical School
Susan B. Meister Child Health Evaluation and Research Center
University of Michigan
MedicalResearch.com: What is the background for this study?
Response: Due to high and rising prices, insulin has become increasingly unaffordable for patients with type 1 diabetes who must pay out-of-pocket for this life-saving medication. Over the past 5 months, many states and insurers have taken steps to cap insulin out-of-pocket spending. For example, Cigna imposed a $25 monthly cap earlier this year. This week, the Centers for Medicare and Medicare Services announced a $35 monthly cap for many Medicare Part D beneficiaries.
Dr. Kao-Ping Chua[/caption]
Kao-Ping Chua, M.D., Ph.D.
Assistant Professor, Pediatrics, Medical School
Susan B. Meister Child Health Evaluation and Research Center
University of Michigan
MedicalResearch.com: What is the background for this study?
Response: Due to high and rising prices, insulin has become increasingly unaffordable for patients with type 1 diabetes who must pay out-of-pocket for this life-saving medication. Over the past 5 months, many states and insurers have taken steps to cap insulin out-of-pocket spending. For example, Cigna imposed a $25 monthly cap earlier this year. This week, the Centers for Medicare and Medicare Services announced a $35 monthly cap for many Medicare Part D beneficiaries.
 Dr. Meiri[/caption]
Amir Meiri, MD MPH
Atrius Health/Department of Population Medicine (DPM) | Delivery System Science Fellow
HMS and HPHCI, DPM | General Internal Medicine Fellow
Atrius Health Kenmore | Urgent Care Physician
VA Boston Healthcare | Attending in Internal Medicine and Emergency Medicine
MedicalResearch.com: What is the background for this study?
Response: There has been significant media reporting about rising insulin prices and the health impacts of those exorbitant prices. However, it was not clear how these insulin prices may impact out-of-pocket costs among commercially insured patients; though it is clear that those without insurance are affected per previous media reports. Our study examines the difference between insulin manufacturer-set prices and what patients actually pay, the out-of-pocket cost, in the context of the type of insurance patients have.
Dr. Meiri[/caption]
Amir Meiri, MD MPH
Atrius Health/Department of Population Medicine (DPM) | Delivery System Science Fellow
HMS and HPHCI, DPM | General Internal Medicine Fellow
Atrius Health Kenmore | Urgent Care Physician
VA Boston Healthcare | Attending in Internal Medicine and Emergency Medicine
MedicalResearch.com: What is the background for this study?
Response: There has been significant media reporting about rising insulin prices and the health impacts of those exorbitant prices. However, it was not clear how these insulin prices may impact out-of-pocket costs among commercially insured patients; though it is clear that those without insurance are affected per previous media reports. Our study examines the difference between insulin manufacturer-set prices and what patients actually pay, the out-of-pocket cost, in the context of the type of insurance patients have.


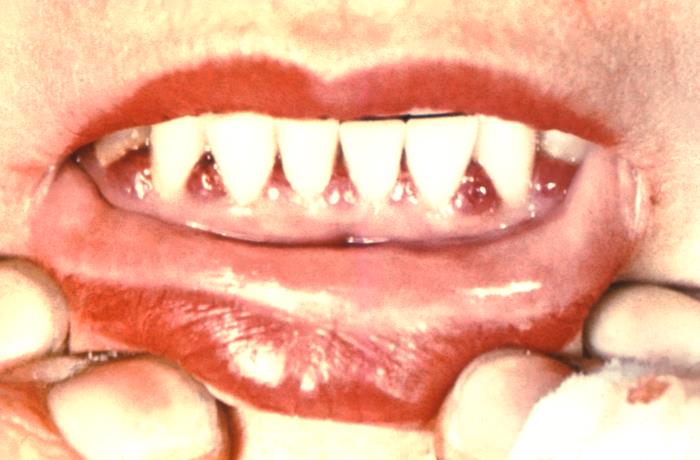
 CDC PHIL image[/caption]
MedicalResearch.com: What is the background for this study?
Response: This report reviewed three measures of tooth loss among adults over 50 years old with selected chronic conditions. The report analyzed data from the 2011–2016 cycles of the National Health and Nutrition Examination Survey (NHANES) to compare the rates of total tooth loss, severe tooth loss (less than 8 teeth), and lacking functional dentition (less than 20 teeth) among people with and without selected chronic conditions.
CDC PHIL image[/caption]
MedicalResearch.com: What is the background for this study?
Response: This report reviewed three measures of tooth loss among adults over 50 years old with selected chronic conditions. The report analyzed data from the 2011–2016 cycles of the National Health and Nutrition Examination Survey (NHANES) to compare the rates of total tooth loss, severe tooth loss (less than 8 teeth), and lacking functional dentition (less than 20 teeth) among people with and without selected chronic conditions.
