Author Interviews, Lung Cancer / 02.05.2018
US vs England: Where Do Lung Cancer Patients Live Longer?
MedicalResearch.com Interview with:
[caption id="attachment_41398" align="alignleft" width="200"]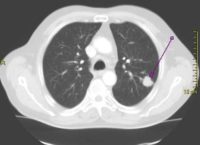 CT scan showing a cancerous tumor in the left lung
CT scan showing a cancerous tumor in the left lung
Wikipedia image[/caption] Cary P. Gross, MD Department of Internal Medicine Section of General Internal Medicine Cancer Outcomes, Public Policy, and Effectiveness Research (COPPER) Center National Clinician Scholars Program Yale School of Medicine New Haven, CT MedicalResearch.com: What is the background for this study? What are the main findings? Response: In both the US and England, lung cancer is a leading cause of cancer deaths, and there is particular concern about access to high quality care among older persons in both countries. However, little is known about how the two nations compare regarding lung cancer care. We studied over 170,000 patients with lung cancer, and found that patients in the US were more likely to be diagnosed at an early stage (25% in US vs 15% of patients in England). Our international team also found that patients in the US were more likely to receive treatment for their cancer, and were more likely to survive.
 CT scan showing a cancerous tumor in the left lung
CT scan showing a cancerous tumor in the left lungWikipedia image[/caption] Cary P. Gross, MD Department of Internal Medicine Section of General Internal Medicine Cancer Outcomes, Public Policy, and Effectiveness Research (COPPER) Center National Clinician Scholars Program Yale School of Medicine New Haven, CT MedicalResearch.com: What is the background for this study? What are the main findings? Response: In both the US and England, lung cancer is a leading cause of cancer deaths, and there is particular concern about access to high quality care among older persons in both countries. However, little is known about how the two nations compare regarding lung cancer care. We studied over 170,000 patients with lung cancer, and found that patients in the US were more likely to be diagnosed at an early stage (25% in US vs 15% of patients in England). Our international team also found that patients in the US were more likely to receive treatment for their cancer, and were more likely to survive.