MedicalResearch.com Interview with:
[caption id="attachment_43604" align="alignleft" width="128"]

Dr. Marcogliese[/caption]
Paul C Marcogliese, Ph.D.
Postdoctoral Associate,
Laboratory of Dr. Hugo Bellen
Department of Molecular and Human Genetics
Baylor College of Medicine
Houston, Texas 77030
[caption id="attachment_43603" align="alignleft" width="99"]

Dr. Peña[/caption]
Loren D. Pena, MD PhD
Division of Human Genetics
Cincinnati Children's Hospital Medical Center
Department of Pediatrics
University of Cincinnati
Cincinnati, OH 45229
MedicalResearch.com: What is the background for this study? What are the main findings?
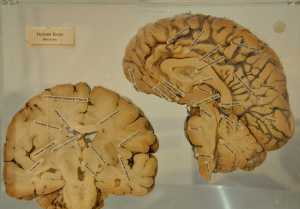
Response: The Undiagnosed Diseases Network (UDN) is a multi-site collaboration across the US that seeks to help diagnose patients with rare disorders that are ill-defined.
Dr. Loren D.M. Pena and Dr. Vandana Shashi at the Duke-Columbia clinical site of the UDN had seen a patient with a severe neurological disorder. While the patient had no symptoms at birth, the patient began falling at about 3 years of age, eventually losing motor coordination and developing seizures. In the interim, the regression has progressed to a severely debilitating state. Re-analysis of the participant’s exome data by our site bioinformatician at Columbia (Nicholas Stong) in Dr. David Goldstein’s laboratory revealed a truncating variant in the single exon gene
IRF2BPL that could be the candidate disease-causing gene. The UDN clinicians at Duke then contacted the UDN Model Organism Screening Center (MOSC) led by Dr. Hugo Bellen at Baylor College of Medicine and the Howard Hughes Medical Institute for functional analysis. In parallel, four more patients were found with truncating mutations causing a similar disorder though the UDN and GeneMatcher.org. Additionally, two patients with missense variants in
IRF2BPL were identified that displayed seizures and some developmental delay or autism spectrum disorder but no motor regression.
Work in MOSC by Dr. Paul Marcogliese using fruit flies revealed that the
IRF2BPL truncating variants are severe loss of function mutations and one of the missense variants was a partial loss of function. Additionally, it was found that the fruit fly
IRF2BPL gene, called
pits, is expressed in the neurons of the adult fly brain. Lowering the levels of
pits by about 50% in fly neurons leads to progressive behavioural abnormalities and neurodegeneration. By combining the human genetics, bioinformatics and model organism data,
IRF2BPL was found to be a novel disease-causing gene in humans.