Author Interviews, Cancer Research, Gastrointestinal Disease, Genetic Research / 03.05.2021
Crohn’s Disease: Genetic and Cellular Mechanisms Identified
MedicalResearch.com Interview with:
[caption id="attachment_57297" align="alignleft" width="114"] Dr. Cho[/caption]
Judy H. Cho, MD,
Dean of Translational Genetics
Director of The Charles Bronfman Institute for Personalized Medicine
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? Would you briefly describe Crohn's disease? Whom does it primarily affect?
Response: Crohn’s disease is a chronic inflammatory intestinal disease, which affects ~3 million Americans a year. Its most typical age of onset ranges from 15-30 years, and many of those diagnosed also exhibit frequent abnormal healing and complications that constrict the digestive tract. The highest risk genetic mutations that increase risk for Crohn’s disease are found in the gene NOD2; these were first reported 20 years ago. Biological mechanisms by which NOD2 mutations drive Crohn’s disease, and especially fibrotic complications, have been incompletely described up until this point. Further, the reasons why many patients fail to respond to the commonly administered anti-TNF treatments also remain incompletely understood.
Dr. Cho[/caption]
Judy H. Cho, MD,
Dean of Translational Genetics
Director of The Charles Bronfman Institute for Personalized Medicine
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? Would you briefly describe Crohn's disease? Whom does it primarily affect?
Response: Crohn’s disease is a chronic inflammatory intestinal disease, which affects ~3 million Americans a year. Its most typical age of onset ranges from 15-30 years, and many of those diagnosed also exhibit frequent abnormal healing and complications that constrict the digestive tract. The highest risk genetic mutations that increase risk for Crohn’s disease are found in the gene NOD2; these were first reported 20 years ago. Biological mechanisms by which NOD2 mutations drive Crohn’s disease, and especially fibrotic complications, have been incompletely described up until this point. Further, the reasons why many patients fail to respond to the commonly administered anti-TNF treatments also remain incompletely understood.
 Dr. Cho[/caption]
Judy H. Cho, MD,
Dean of Translational Genetics
Director of The Charles Bronfman Institute for Personalized Medicine
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? Would you briefly describe Crohn's disease? Whom does it primarily affect?
Response: Crohn’s disease is a chronic inflammatory intestinal disease, which affects ~3 million Americans a year. Its most typical age of onset ranges from 15-30 years, and many of those diagnosed also exhibit frequent abnormal healing and complications that constrict the digestive tract. The highest risk genetic mutations that increase risk for Crohn’s disease are found in the gene NOD2; these were first reported 20 years ago. Biological mechanisms by which NOD2 mutations drive Crohn’s disease, and especially fibrotic complications, have been incompletely described up until this point. Further, the reasons why many patients fail to respond to the commonly administered anti-TNF treatments also remain incompletely understood.
Dr. Cho[/caption]
Judy H. Cho, MD,
Dean of Translational Genetics
Director of The Charles Bronfman Institute for Personalized Medicine
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? Would you briefly describe Crohn's disease? Whom does it primarily affect?
Response: Crohn’s disease is a chronic inflammatory intestinal disease, which affects ~3 million Americans a year. Its most typical age of onset ranges from 15-30 years, and many of those diagnosed also exhibit frequent abnormal healing and complications that constrict the digestive tract. The highest risk genetic mutations that increase risk for Crohn’s disease are found in the gene NOD2; these were first reported 20 years ago. Biological mechanisms by which NOD2 mutations drive Crohn’s disease, and especially fibrotic complications, have been incompletely described up until this point. Further, the reasons why many patients fail to respond to the commonly administered anti-TNF treatments also remain incompletely understood.

 Dr. Ulhas Nair[/caption]
Nishanth Ulhas Nair, Ph.D.
Affiliation: Staff Scientist at Cancer Data Science Laboratory, Center for Cancer Research
National Cancer Institute (NCI), National Institutes of Health (NIH)
Bethesda, Maryland, USA. Date: April 22, 2021
Dr. Raffit Hassan and Dr. Eytan Ruppin at the National Cancer Institute (NCI) are the senior authors of this study.
MedicalResearch.com: What is the background for this study?
Response: Malignant mesothelioma is an aggressive cancer with limited treatment options and poor prognosis. An in-depth knowledge of genetic, transcriptomic and immunogenic events involved in mesothelioma is critical for successful development of prognostics and therapeutic modalities. In this study we aim to address this by exploring a new large scale patient tumor dataset of 122 mesothelioma patients, called NCI mesothelioma patient data, along with their genomic, transcriptomic, and phenotypic information. Unlike previous large-scale studies which have been focused on malignant pleural mesothelioma patients, our dataset contains an approximately equal representation of malignant pleural and peritoneal mesothelioma patients which allows to identify any differences between them.
Dr. Ulhas Nair[/caption]
Nishanth Ulhas Nair, Ph.D.
Affiliation: Staff Scientist at Cancer Data Science Laboratory, Center for Cancer Research
National Cancer Institute (NCI), National Institutes of Health (NIH)
Bethesda, Maryland, USA. Date: April 22, 2021
Dr. Raffit Hassan and Dr. Eytan Ruppin at the National Cancer Institute (NCI) are the senior authors of this study.
MedicalResearch.com: What is the background for this study?
Response: Malignant mesothelioma is an aggressive cancer with limited treatment options and poor prognosis. An in-depth knowledge of genetic, transcriptomic and immunogenic events involved in mesothelioma is critical for successful development of prognostics and therapeutic modalities. In this study we aim to address this by exploring a new large scale patient tumor dataset of 122 mesothelioma patients, called NCI mesothelioma patient data, along with their genomic, transcriptomic, and phenotypic information. Unlike previous large-scale studies which have been focused on malignant pleural mesothelioma patients, our dataset contains an approximately equal representation of malignant pleural and peritoneal mesothelioma patients which allows to identify any differences between them.







 Dr. Magnusson[/caption]
Karin Magnusson PT, PhD
Associate Researcher
Lund University and
Norwegian Institute of Public Health
MedicalResearch.com: What is the background for this study?
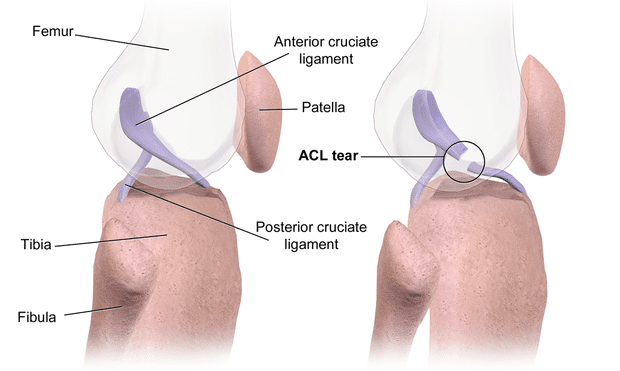
Response: Anterior cruciate ligament (ACL) injury is one of the most common knee injuries, for which very limited data has been presented on the genetic contribution. Based on our knowledge of the role of genetics in the development of ACL-rupture related traits, such as joint hypermobility and knee osteoarthritis, we hypothesized that heritability might play a role also in ACL injury.
Using the Swedish Twin Registry, which is the world's largest twin registry and in this study including more than 88.000 twins, we had unique data to for the first time reliably estimate the heritability for this common knee injury.
Dr. Magnusson[/caption]
Karin Magnusson PT, PhD
Associate Researcher
Lund University and
Norwegian Institute of Public Health
MedicalResearch.com: What is the background for this study?
Response: Anterior cruciate ligament (ACL) injury is one of the most common knee injuries, for which very limited data has been presented on the genetic contribution. Based on our knowledge of the role of genetics in the development of ACL-rupture related traits, such as joint hypermobility and knee osteoarthritis, we hypothesized that heritability might play a role also in ACL injury.
Using the Swedish Twin Registry, which is the world's largest twin registry and in this study including more than 88.000 twins, we had unique data to for the first time reliably estimate the heritability for this common knee injury. 
 Dr. Willette[/caption]
Auriel Willette, PhD
Assistant Professor
Food Science and Human Nutrition
Iowa State University
MedicalResearch.com: What is the background for this study?
Response: To date, pharmacology therapies done to slow down or halt Alzheimer's disease have been inconclusive. Lifestyle interventions like changes in diet and activity are also mixed but do show some promise. Dietary clinical trials or self-reported diet have tended to focus on groups of foods such as the Mediterranean or MIND diet. To build from this excellent work, we were curious if we could pinpoint specific foods that were correlated with changes in fluid intelligence over time. Fluid intelligence represents our ability to creatively use existing knowledge, working memory, and other components of "thinking flexibly."
Further, we tested if these patterns of association differed based on genetic risk. In this case, genetic risk was defined as having a family history of Alzheimer's disease or having 1-2 "bad" copies of the Apolipoprotein E (APOE) gene, which is the strongest genetic risk factor for Alzheimer's disease.
Dr. Willette[/caption]
Auriel Willette, PhD
Assistant Professor
Food Science and Human Nutrition
Iowa State University
MedicalResearch.com: What is the background for this study?
Response: To date, pharmacology therapies done to slow down or halt Alzheimer's disease have been inconclusive. Lifestyle interventions like changes in diet and activity are also mixed but do show some promise. Dietary clinical trials or self-reported diet have tended to focus on groups of foods such as the Mediterranean or MIND diet. To build from this excellent work, we were curious if we could pinpoint specific foods that were correlated with changes in fluid intelligence over time. Fluid intelligence represents our ability to creatively use existing knowledge, working memory, and other components of "thinking flexibly."
Further, we tested if these patterns of association differed based on genetic risk. In this case, genetic risk was defined as having a family history of Alzheimer's disease or having 1-2 "bad" copies of the Apolipoprotein E (APOE) gene, which is the strongest genetic risk factor for Alzheimer's disease.











 Prof. Medland[/caption]
Professor Sarah Medland
Coordinator of the Mental Health Research Program and Group Leader Psychiatric Genetics
QIMR Berghofer Medical Research Institute
MedicalResearch.com: What is the background for this study?
Response: This large collaborative project involving participants and researchers from around the world which has been underway for about 10 years. The aim was to try and identify genetic variants that influence handedness with the goal of increasing our knowledge about the way lateralization develops in behaviour and in the brain.
In this project we were able to bring together results from cohort studies conducted by academic collaborators, the UK Biobank and 23andMe yielding a total sample size of over 1.7 million participants. Working with Professor David Evans the co-senior author of the paper (University of Queensland) and Dr Gabriel Cuellar-Partida the first author of the paper (formally UQ now at 23andMe) and the other researchers who worked on the project we meta-analysed the genome-wide association analysis results from the cohorts and were able to identify 41 genetic variants that influence left-handedness and 7 that influence ambidextrousness.
Prof. Medland[/caption]
Professor Sarah Medland
Coordinator of the Mental Health Research Program and Group Leader Psychiatric Genetics
QIMR Berghofer Medical Research Institute
MedicalResearch.com: What is the background for this study?
Response: This large collaborative project involving participants and researchers from around the world which has been underway for about 10 years. The aim was to try and identify genetic variants that influence handedness with the goal of increasing our knowledge about the way lateralization develops in behaviour and in the brain.
In this project we were able to bring together results from cohort studies conducted by academic collaborators, the UK Biobank and 23andMe yielding a total sample size of over 1.7 million participants. Working with Professor David Evans the co-senior author of the paper (University of Queensland) and Dr Gabriel Cuellar-Partida the first author of the paper (formally UQ now at 23andMe) and the other researchers who worked on the project we meta-analysed the genome-wide association analysis results from the cohorts and were able to identify 41 genetic variants that influence left-handedness and 7 that influence ambidextrousness.






