Author Interviews, Gender Differences, Hepatitis - Liver Disease, Infections / 10.07.2020
Cost-Effectiveness of Routine Hepatitis C Testing
MedicalResearch.com Interview with:
[caption id="attachment_54838" align="alignleft" width="150"] Dr. Assoumou[/caption]
Sabrina Annick Assoumou, MD, MPH
Assistant Professor, Medicine
Infectious Diseases at Boston Medical Center
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: During the opioid epidemic there has been an increase in the number of hepatitis C virus (HCV) infections due to transmission among persons who inject drugs (PWID). Federally qualified health centers (FQHC) provide care to an underserved and diverse patient population with a high proportion of both injection drug use and HCV. These health care facilities could provide opportunities to enhance HCV testing and treatment, especially at a time when recent data show that the United States is not on the list of high-income nations expected to achieve the World Health Organization’s goal of eliminating HCV by 2030.
Dr. Assoumou[/caption]
Sabrina Annick Assoumou, MD, MPH
Assistant Professor, Medicine
Infectious Diseases at Boston Medical Center
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: During the opioid epidemic there has been an increase in the number of hepatitis C virus (HCV) infections due to transmission among persons who inject drugs (PWID). Federally qualified health centers (FQHC) provide care to an underserved and diverse patient population with a high proportion of both injection drug use and HCV. These health care facilities could provide opportunities to enhance HCV testing and treatment, especially at a time when recent data show that the United States is not on the list of high-income nations expected to achieve the World Health Organization’s goal of eliminating HCV by 2030.
 Dr. Assoumou[/caption]
Sabrina Annick Assoumou, MD, MPH
Assistant Professor, Medicine
Infectious Diseases at Boston Medical Center
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: During the opioid epidemic there has been an increase in the number of hepatitis C virus (HCV) infections due to transmission among persons who inject drugs (PWID). Federally qualified health centers (FQHC) provide care to an underserved and diverse patient population with a high proportion of both injection drug use and HCV. These health care facilities could provide opportunities to enhance HCV testing and treatment, especially at a time when recent data show that the United States is not on the list of high-income nations expected to achieve the World Health Organization’s goal of eliminating HCV by 2030.
Dr. Assoumou[/caption]
Sabrina Annick Assoumou, MD, MPH
Assistant Professor, Medicine
Infectious Diseases at Boston Medical Center
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: During the opioid epidemic there has been an increase in the number of hepatitis C virus (HCV) infections due to transmission among persons who inject drugs (PWID). Federally qualified health centers (FQHC) provide care to an underserved and diverse patient population with a high proportion of both injection drug use and HCV. These health care facilities could provide opportunities to enhance HCV testing and treatment, especially at a time when recent data show that the United States is not on the list of high-income nations expected to achieve the World Health Organization’s goal of eliminating HCV by 2030.

 Dr. Dixon[/caption]
Cinnamon A. Dixon, DO, MPH
Associate Professor of Pediatrics
University of Colorado School of Medicine
Children’s Hospital Colorado
Senior Investigator | Center for Global Health
Colorado School of Public Health
Aurora, CO
MedicalResearch.com: What is the background for this commentary?
Response: Dog bites are a long-standing public health problem. Each year there are approximately 4.5 million dog bites across the Unites States (US),1 and global estimates suggest tens of millions of these injuries worldwide.2 Children are the most vulnerable population with nearly 1 million annual dog bites in the US and more severe injury outcomes.1
National organizations espouse consistent strategies on how to prevent dog bites to children, however studies reveal that most children have never received dog bite prevention education.3,4 Furthermore, children lack critical knowledge of how to prevent dog bites in high-risk “resource guarding” situations (such as when a dog is eating or chewing on toys).4
During the COVID-19 pandemic, millions of US households are experiencing restrictions in activities. Children now spend more time in the home environment and presumably have increased exposure to their pet dogs. Parents and caregivers likely experience greater stress with more potential for competing interests and resultant decreased supervision of their children and dogs. Finally, pet dogs may be affected by the increased tension of their environment and be more likely to mirror the emotions of their human caregivers.
We hypothesized that these combined elements compound the risk of dog bites to children during the COVID-19 pandemic.
Dr. Dixon[/caption]
Cinnamon A. Dixon, DO, MPH
Associate Professor of Pediatrics
University of Colorado School of Medicine
Children’s Hospital Colorado
Senior Investigator | Center for Global Health
Colorado School of Public Health
Aurora, CO
MedicalResearch.com: What is the background for this commentary?
Response: Dog bites are a long-standing public health problem. Each year there are approximately 4.5 million dog bites across the Unites States (US),1 and global estimates suggest tens of millions of these injuries worldwide.2 Children are the most vulnerable population with nearly 1 million annual dog bites in the US and more severe injury outcomes.1
National organizations espouse consistent strategies on how to prevent dog bites to children, however studies reveal that most children have never received dog bite prevention education.3,4 Furthermore, children lack critical knowledge of how to prevent dog bites in high-risk “resource guarding” situations (such as when a dog is eating or chewing on toys).4
During the COVID-19 pandemic, millions of US households are experiencing restrictions in activities. Children now spend more time in the home environment and presumably have increased exposure to their pet dogs. Parents and caregivers likely experience greater stress with more potential for competing interests and resultant decreased supervision of their children and dogs. Finally, pet dogs may be affected by the increased tension of their environment and be more likely to mirror the emotions of their human caregivers.
We hypothesized that these combined elements compound the risk of dog bites to children during the COVID-19 pandemic.



 Samia Arshad[/caption]
Samia Arshad, MPH
Epidemiologist II
Infectious Disease
Henry Ford Hospital, Detroit, MI
I would like to start off by saying: We need to keep partisanship out of science. During this pandemic, we hope we can stick to science and help save lives with purposeful data driven facts.
MedicalResearch.com: What is the background for this study?
Response: Hydroxychloroquine, an antimalarial and immunomodulatory agent has demonstrated antiviral activity against SARS-CoV-2. We are in an acceleration phase of the COVID-19 pandemic, with 25% of the world’s cases occurring in the United States. Currently there is no known therapy or vaccine for treatment of SARS-CoV-2, highlighting the urgency around identifying effective therapies. The purpose of this study was to evaluate the role of hydroxychloroquine therapy alone and in combination with azithromycin in hospitalized patients positive for COVID-19.
Samia Arshad[/caption]
Samia Arshad, MPH
Epidemiologist II
Infectious Disease
Henry Ford Hospital, Detroit, MI
I would like to start off by saying: We need to keep partisanship out of science. During this pandemic, we hope we can stick to science and help save lives with purposeful data driven facts.
MedicalResearch.com: What is the background for this study?
Response: Hydroxychloroquine, an antimalarial and immunomodulatory agent has demonstrated antiviral activity against SARS-CoV-2. We are in an acceleration phase of the COVID-19 pandemic, with 25% of the world’s cases occurring in the United States. Currently there is no known therapy or vaccine for treatment of SARS-CoV-2, highlighting the urgency around identifying effective therapies. The purpose of this study was to evaluate the role of hydroxychloroquine therapy alone and in combination with azithromycin in hospitalized patients positive for COVID-19. ![MedicalResearch.com Interview with: Dr Herman Anne Service de Dermatologie Cliniques Universitaires Saint-Luc Avenue Hippocrate, 10 1200 Bruxelle MedicalResearch.com: What is the background for this study? Response: In the context of the COVID-19 pandemic, several cases of acro-located lesions (on foot or hands) suggestive of chilblains have been reported and were possibly related to COVID-19. We wanted to determine if chilblains, observed in many patients recently referred to our department, are indicative of COVID-19. MedicalResearch.com: Would you briefly explain what is meant by chilblains? Response: Chilblains are frequent cold induced inflammatory lesions. Chilblains are typically seen in winter and occur after repeated exposure to cold temperatures. Clinical presentation includes erythema and swelling on toes and/or digits followed by red-purple macules or patches. However, given the large number of patients affected, and the exceptionally high outdoor temperatures for the spring season over the past month and at the time of case-observation, cold-exposure seemed unlikely. These lesions were, therefore, suspected to be associated with COVID-19. However, to date, no study has proven a pathological link between these lesions and COVID-19. MedicalResearch.com: What are the main findings? Response: In our series of 31 patients who recently developed chilblains, negative nasopharyngeal swabs and the absence of anti-SARS-CoV-2 blood immunoglobulin (Ig)M and IgG antibodies in all patients included in your study suggested that these patients had not been infected with COVID-19. These lesions appeared not to be directly related to COVID-19. MedicalResearch.com: What should readers take away from your report? Response: One hypothesis points to an indirect consequence of the COVID-19 pandemic due to imposed community containment and lockdown measures. Indeed, all patients in this study had either been working from home or were home-schooled since the beginning of containment measures in Belgium (March 11, 2020) or were temporarily unemployed. As a result of containment measures, the majority (64.5%) of patients reported a decrease in their physical activity and significantly more time spent in sedentary positions in front of screens. Most of the patients declared that they remained barefoot or in socks most of the day. All these lifestyle changes can be considered as risk factors for developing chilblains. Therefore, it seems plausible that containment, through its collateral effects, may induce chilblains. Interestingly also, the mean BMI of the patients included was relatively low, suggesting that thin people may be more at risk of developing chilblains. MedicalResearch.com: What recommendations do you have for future research as a result of this work? Response: During the current pandemic, several reports have suggested a possible link between cutaneous manifestations including aural lesions such as chilblains, and COVID-19, however, only few patients were tested for SARS-CoV-2 by RT-PCR and no serologic tests were performed. Therefore, reliable testings (RT-PCR and serologic testing) are essential to confirm a potential association with COVID-19. Citation: Herman A, Peeters C, Verroken A, et al. Evaluation of Chilblains as a Manifestation of the COVID-19 Pandemic. JAMA Dermatol. Published online June 25, 2020. doi:10.1001/jamadermatol.2020.2368 https://jamanetwork.com/journals/jamadermatology/fullarticle/2767774 [subscribe] Last Modified: [last-modified] The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2020/06/chilblain-example-dermnet-nz.jpg)

 Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination
Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination 
 Dr. Blume[/caption]
Dr. Christine Blume PhD
Centre for Chronobiology
Psychiatric Hospital of the University of Basel
Transfaculty Research Platform Molecular and Cognitive Neurosciences
Basel
MedicalResearch.com: What is the background for this study?
Response: In modern societies, human rest-activity rhythms and sleep are between the often-conflicting poles of external social time (e.g., work hours and leisure activities) and an individual’s internal biological time. This can lead to so-called “social jetlag”, which has repeatedly been associated with detrimental health effects. With the restrictions to control the pandemic, social timing relaxed as people many started working from home and public life came to a standstill. In an online survey with 435 respondents, we investigated the effects of the phase with the strictest COVID-19 restrictions on the relationship between social and biological rhythms as well as sleep during a six-week period (mid-March until end of April 2020) in three European societies (Austria, Germany, Switzerland).
Dr. Blume[/caption]
Dr. Christine Blume PhD
Centre for Chronobiology
Psychiatric Hospital of the University of Basel
Transfaculty Research Platform Molecular and Cognitive Neurosciences
Basel
MedicalResearch.com: What is the background for this study?
Response: In modern societies, human rest-activity rhythms and sleep are between the often-conflicting poles of external social time (e.g., work hours and leisure activities) and an individual’s internal biological time. This can lead to so-called “social jetlag”, which has repeatedly been associated with detrimental health effects. With the restrictions to control the pandemic, social timing relaxed as people many started working from home and public life came to a standstill. In an online survey with 435 respondents, we investigated the effects of the phase with the strictest COVID-19 restrictions on the relationship between social and biological rhythms as well as sleep during a six-week period (mid-March until end of April 2020) in three European societies (Austria, Germany, Switzerland).
![MedicalResearch.com Interview with: Sarah Messiah, PhD, MPH Professor of epidemiology, human genetics, and environmental sciences UTHealth School of Public Health Dallas MedicalResearch.com: What is the background for this study? Response: Given that obesity is a prevalent, serious, complex, chronic, and relapsing disease, and severe obesity is a deepening crisis, it is important to pay special attention to these challenges during the COVID-19 pandemic. This will avoid placing an even greater burden on individuals, health systems, and society in the post-COVID-19 recovery phase. Thus, it is critically important to document, in real time, how socioecological determinants of health are impacting behaviors among those with obesity. Before our study, how the COVID-19 pandemic is impacting weight management, health behaviors, and psychosocial health in particular among people with obesity was unknown. MedicalResearch.com: What are the main findings? Response: A total of 123 patients (87% female, mean age 51.2 years, mean BMI 40.2, 49.2% Non-Hispanic white, 28.7% Non-Hispanic black, 16.4% Hispanic, 7% other ethnicity, 33.1% completed bariatric surgery were included). -Two patients tested positive for SARS-CoV-2 and 14.6% reported symptoms. -10% lost their job since the beginning of the pandemic -25% reported food insecurity in that they sometimes, or always could not afford to eat balanced meals -72.8% reported increased anxiety and 83.6% increased depression since stay-at-home orders were initiated. -69.6% reported more difficultly in achieving weight loss goals -47.9% reported less exercise time and 55.8% reported less intensity -49.6% reported increased stockpiling of food -61.2% reported stress eating -61.2% reported following healthy diet plans more challenging - Hispanics were less likely to report anxiety versus non-Hispanic whites MedicalResearch.com: What should readers take away from your report? Response: In a relatively short period of time, our results show that there have been substantial perceptions in changes in health behaviors among patients with obesity. Our sample described a decreases in positive health behaviors, increases in deleterious behaviors and associated deterioration in mental health. Even though actual COVID-19 disease burden was low (1.7% tested positive for COVID-19 and another 14.6% reported symptoms), the pandemic is having a significant impact on those without infections. The major strength of this study is that it is the first snapshot into how the COVID-19 pandemic has influenced health behaviors for patients with obesity. Prior to the pandemic, it has been reported that Americans consume 20% of their calories from restaurants and that there are worsening disparities in fast food meal quality by race/ethnicity, education, and income. Due to recent economic challenges, patients may be more likely to select cheaper foods, which are often energy dense and nutrient poor. Therefore, even though we found more patients are cooking at home, the type of foods that are being stockpiled are likely to be processed foods due to their longer shelf life. These are associated with greater intake of fat, carbohydrate, and calories, which facilitate greater weight gain when compared to more balanced diets. It is possible Americans may be trading one pattern of low-quality consumption for another by choosing cheaper processed foods instead of eating out. MedicalResearch.com: What recommendations do you have for future research as a result of this work? Response: These results have implications for clinical practice and management of patients with obesity as we now move into post-COVID-19 relief efforts. Due to the increase in obesogenic behaviors related to the COVID-19 pandemic that were found here, it is paramount that healthcare access is not disrupted for patients with obesity. Maintaining these vital services will prevent exacerbating the negative health and economic consequences of excess body weight. This includes access to primary care providers, Obesity Medicine specialists and bariatric surgery programs. In addition to asking about diet and exercise patterns, screening for indicators of mental health, loneliness, financial stressors and behaviors that may influence body weight should be implemented by healthcare teams to combat this problem. Disclosures: This work was funded by the National Institutes of Health, National Institute on Minority Health and Health Disparities (Grant #R01MD011686). This work was a joint collaboration among investigators from UTHealth School of Public Health, UT Southwestern Medical Center and Minimally Invasive Surgical Associates. Citation: Almandoz, J.P., Xie, L., Schellinger, J.N., Mathew, M.S., Gazda, C., Ofori, A., Kukreja, S. and Messiah, S.E. (2020), Impact of COVID ‐19 Stay‐at‐Home Orders on Weight‐Related Behaviors Among Patients with Obesity. Clin Obes. Accepted Author Manuscript. doi:10.1111/cob.12386 [subscribe] [last-modified] The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2020/06/Dr-Sarah-E.-Messiah.jpg)

 Dr. Hooker[/caption]
Edmond A. Hooker, MD, DrPH
Professor, Department of Health Services Administration
MHSA Associate Director for Accreditation
Xavier University
Cincinnati, Ohio
MedicalResearch.com: What is the background for this study?
Response: Healthcare acquired infections are still a huge problem in the US. Most hospitals are not following the hospital bed manufacturers guidelines for cleaning these mattresses and bed decks (the metal bed frame). Many use only a single step, when the manufacturers recommend a 5-6 step process (pre-clean any obvious soil, clean, rinse off cleaner, disinfect, rinse off disinfectant, inspect mattress for damage). The problem is that, if done, this takes 45 minutes to 1 hours. Most hospitals turn over a hospital room in less than 30 minutes. The launderable bed cover allows the bed to be cleaned and turned over in minutes, not an hours.
Dr. Hooker[/caption]
Edmond A. Hooker, MD, DrPH
Professor, Department of Health Services Administration
MHSA Associate Director for Accreditation
Xavier University
Cincinnati, Ohio
MedicalResearch.com: What is the background for this study?
Response: Healthcare acquired infections are still a huge problem in the US. Most hospitals are not following the hospital bed manufacturers guidelines for cleaning these mattresses and bed decks (the metal bed frame). Many use only a single step, when the manufacturers recommend a 5-6 step process (pre-clean any obvious soil, clean, rinse off cleaner, disinfect, rinse off disinfectant, inspect mattress for damage). The problem is that, if done, this takes 45 minutes to 1 hours. Most hospitals turn over a hospital room in less than 30 minutes. The launderable bed cover allows the bed to be cleaned and turned over in minutes, not an hours. 



 Dr. Schünemann[/caption]
Holger Schünemann, MD, PhD, FRCPC
Professor of Clinical Epidemiology and of Medicine
Co-Director, WHO Collaborating Centre for Infectious Diseases,
Research Methods and Recommendations
Director, Cochrane Canada and McMaster GRADE Centre
Department of Health Research Methods, Evidence, and Impact
Canada
MedicalResearch.com: What is the background for this study?
Response: Many countries and regions have issued conflicting advice about physical distancing to reduce transmission of COVID-19, based on limited information. In addition, the questions of whether masks and eye coverings might reduce transmission of COVID-19 in the general population, and what the optimum use of masks in healthcare settings is, have been debated during the pandemic.
Dr. Schünemann[/caption]
Holger Schünemann, MD, PhD, FRCPC
Professor of Clinical Epidemiology and of Medicine
Co-Director, WHO Collaborating Centre for Infectious Diseases,
Research Methods and Recommendations
Director, Cochrane Canada and McMaster GRADE Centre
Department of Health Research Methods, Evidence, and Impact
Canada
MedicalResearch.com: What is the background for this study?
Response: Many countries and regions have issued conflicting advice about physical distancing to reduce transmission of COVID-19, based on limited information. In addition, the questions of whether masks and eye coverings might reduce transmission of COVID-19 in the general population, and what the optimum use of masks in healthcare settings is, have been debated during the pandemic.







 Dr. Van der Pol[/caption]
Barbara Van Der Pol, PhD, MPH
President, American STD Association
President-Elect, International Society fo STD Research
Professor of Medicine & Public Health
Director, STD Diagnostics Lab
Director, UAB STD Clinical Research Organization
University of Alabama at Birmingham
Birmingham, AL 35294
MedicalResearch.com: What is the background for this study?
Response: For many decades, public health programs focused on control and prevention of sexually transmitted infections (STI) have relied on lab based tests that required a 1-day to 1-week wait for test results. Efforts to shorten the duration of infection lead to treating at the time of the clinic visit based on the association between certain clinical signs or symptoms and the likelihood of infection.
Unfortunately, more than ½ of infected persons do not have signs or symptoms so they receive no treatment until test results are available. Thus, a test that could be performed at the clinical site with a limited wait time would improve the accuracy of treatment and shorten the duration of infection. Such a strategy will hopefully reduce the number of return clinic visits, the possibility of transmission, and the potential for consequences of untreated infection such as infertility.
Dr. Van der Pol[/caption]
Barbara Van Der Pol, PhD, MPH
President, American STD Association
President-Elect, International Society fo STD Research
Professor of Medicine & Public Health
Director, STD Diagnostics Lab
Director, UAB STD Clinical Research Organization
University of Alabama at Birmingham
Birmingham, AL 35294
MedicalResearch.com: What is the background for this study?
Response: For many decades, public health programs focused on control and prevention of sexually transmitted infections (STI) have relied on lab based tests that required a 1-day to 1-week wait for test results. Efforts to shorten the duration of infection lead to treating at the time of the clinic visit based on the association between certain clinical signs or symptoms and the likelihood of infection.
Unfortunately, more than ½ of infected persons do not have signs or symptoms so they receive no treatment until test results are available. Thus, a test that could be performed at the clinical site with a limited wait time would improve the accuracy of treatment and shorten the duration of infection. Such a strategy will hopefully reduce the number of return clinic visits, the possibility of transmission, and the potential for consequences of untreated infection such as infertility. 
 Marco Taglietti, M.D.
President and Chief Executive Officer
SCYNEXIS Inc
Dr. Taglietti discusses SCYNEXIS’ announcement of positive results from its second Phase 3 study investigating the safety and efficacy of oral ibrexafungerp as a treatment for vaginal yeast infection.
[caption id="attachment_54239" align="alignleft" width="200"]
Marco Taglietti, M.D.
President and Chief Executive Officer
SCYNEXIS Inc
Dr. Taglietti discusses SCYNEXIS’ announcement of positive results from its second Phase 3 study investigating the safety and efficacy of oral ibrexafungerp as a treatment for vaginal yeast infection.
[caption id="attachment_54239" align="alignleft" width="200"]

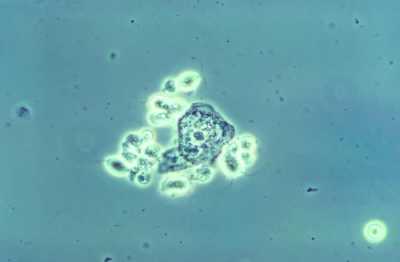
 Dr. Kaufman[/caption]
Gregory Kaufman, M.D.
Senior Vice President
Global Clinical and Medical Affairs
Specialty at Lupin
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by trichomoniasis? How common is this infection?
Response: The Phase 3 trial evaluated the effectiveness and safety of a single oral dose of Solosec® (secnidazole) 2g oral granules for the treatment of trichomoniasis in adult women. Top-line results were positive and showed that Solosec was generally well-tolerated.
Trichomoniasis is the most common non-viral sexually transmitted infection (STI) in the U.S., and is caused by a protozoan parasite called Trichomonas vaginalis.
Dr. Kaufman[/caption]
Gregory Kaufman, M.D.
Senior Vice President
Global Clinical and Medical Affairs
Specialty at Lupin
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by trichomoniasis? How common is this infection?
Response: The Phase 3 trial evaluated the effectiveness and safety of a single oral dose of Solosec® (secnidazole) 2g oral granules for the treatment of trichomoniasis in adult women. Top-line results were positive and showed that Solosec was generally well-tolerated.
Trichomoniasis is the most common non-viral sexually transmitted infection (STI) in the U.S., and is caused by a protozoan parasite called Trichomonas vaginalis.