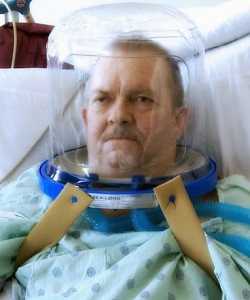
Author Interviews, COVID -19 Coronavirus, Pulmonary Disease / 04.04.2020
Respiratory Helmets Can Decrease Need for Intubation and Spread of COVID-19 Virus
MedicalResearch.com Interview with:
[caption id="attachment_53773" align="alignleft" width="165"] Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.
Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.
 Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.
Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.









 Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.
Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.







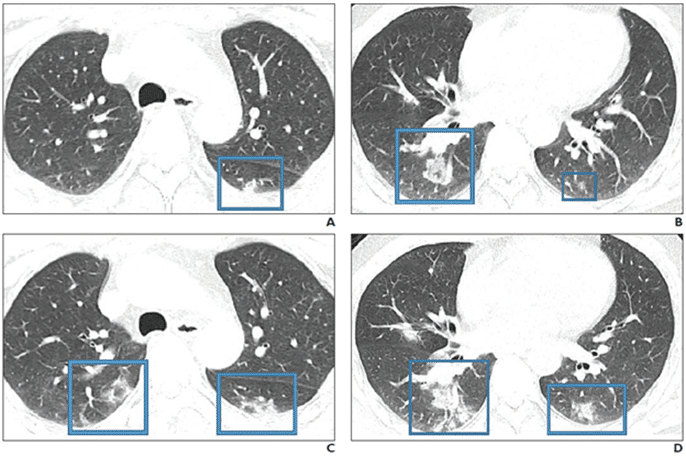
 Dr. Kooraki[/caption]
Soheil Kooraki MSR MS, MD
on behalf of Dr. Ali Gholamrezanezhad MD and co-authors
Department of Radiological Sciences,
David Geffen School of Medicine, University of California at Los Angeles
Los Angeles, California
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: COVID19 is a novel strain of the coronavirus family causing pneumonia. Two similar strains were discovered in 2003 and 2012 to cause the so-called SARS and MERS outbreaks, respectively. Radiologists need to be prepared for the escalating incidence of COVID-19. We reviewed the literature to extract the epidemiologic and imaging features of SARS and MERS in comparison with known imaging features of COVID-19 pneumonia to have a better understanding of the imaging features of the COVID19 pneumonia in acute and post-recovery stages.
Dr. Kooraki[/caption]
Soheil Kooraki MSR MS, MD
on behalf of Dr. Ali Gholamrezanezhad MD and co-authors
Department of Radiological Sciences,
David Geffen School of Medicine, University of California at Los Angeles
Los Angeles, California
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: COVID19 is a novel strain of the coronavirus family causing pneumonia. Two similar strains were discovered in 2003 and 2012 to cause the so-called SARS and MERS outbreaks, respectively. Radiologists need to be prepared for the escalating incidence of COVID-19. We reviewed the literature to extract the epidemiologic and imaging features of SARS and MERS in comparison with known imaging features of COVID-19 pneumonia to have a better understanding of the imaging features of the COVID19 pneumonia in acute and post-recovery stages.




 Dr. Marra[/caption]
Alexandre R. Marra, MD PhD
Iowa Infection Prevention Research Group
University of Iowa Carver College of Medicine
Division of Medical Practice, Hospital Israelita Albert Einstein
São Paulo, Brazil
MedicalResearch.com: What is the background for this study?
Response: This is a comprehensive systematic literature review evaluating the burden of C. difficile infections in the U.S healthcare system. The literature has diverse studies with variable outcomes. Thus, we only included incidence estimates derived from multicenter studies. In our meta-analysis to calculate incidence, data were pooled only with denominators using the same unit (patient-days) to avoid comparisons with different denominators. For length of stay (LOS), we only included studies that used advanced statistical methods (e.g., propensity score matching).
Dr. Marra[/caption]
Alexandre R. Marra, MD PhD
Iowa Infection Prevention Research Group
University of Iowa Carver College of Medicine
Division of Medical Practice, Hospital Israelita Albert Einstein
São Paulo, Brazil
MedicalResearch.com: What is the background for this study?
Response: This is a comprehensive systematic literature review evaluating the burden of C. difficile infections in the U.S healthcare system. The literature has diverse studies with variable outcomes. Thus, we only included incidence estimates derived from multicenter studies. In our meta-analysis to calculate incidence, data were pooled only with denominators using the same unit (patient-days) to avoid comparisons with different denominators. For length of stay (LOS), we only included studies that used advanced statistical methods (e.g., propensity score matching).