Author Interviews, Cancer Research / 17.12.2025
Icahn Mount Sinai Scientists Engineer mRNA to Selectively Target Cancer Cells
[caption id="attachment_71812" align="alignleft" width="200"] Dr. Zak[/caption]
MedicalResearch.com Interview with:
Magdalena M. Żak, PhD
Zangi Lab
Instructor | Cardiovascular Research Institute
Instructor | Genetics & Genomic Sciences
Icahn School of Medicine at Mount Sinai
Hess Center for Science and Medicine
New York, NY 10029
MedicalResearch.com: What is the background for this study?
Response: mRNA has proven to be a groundbreaking technology through COVID-19 vaccines, and most mRNA-based therapeutics in development today are still focused on vaccines. However, in principle, mRNA could be used for many diseases in which expression of a therapeutic protein would be beneficial. A major reason mRNA is less commonly used outside of vaccines is the lack of robust targeting: for vaccination, broad expression can be acceptable because the goal is antigen production for immune recognition, but for other applications - especially cancer - targeted delivery and minimized off-target expression are critical to reduce side effects. Current targeting strategies largely rely on lipid nanoparticles (LNPs), which act as lipid “carriers” for systemic delivery. Although LNPs can be designed to show some tissue tropism, this is often limited to organs such as the liver, spleen, and lungs.
Dr. Zak[/caption]
MedicalResearch.com Interview with:
Magdalena M. Żak, PhD
Zangi Lab
Instructor | Cardiovascular Research Institute
Instructor | Genetics & Genomic Sciences
Icahn School of Medicine at Mount Sinai
Hess Center for Science and Medicine
New York, NY 10029
MedicalResearch.com: What is the background for this study?
Response: mRNA has proven to be a groundbreaking technology through COVID-19 vaccines, and most mRNA-based therapeutics in development today are still focused on vaccines. However, in principle, mRNA could be used for many diseases in which expression of a therapeutic protein would be beneficial. A major reason mRNA is less commonly used outside of vaccines is the lack of robust targeting: for vaccination, broad expression can be acceptable because the goal is antigen production for immune recognition, but for other applications - especially cancer - targeted delivery and minimized off-target expression are critical to reduce side effects. Current targeting strategies largely rely on lipid nanoparticles (LNPs), which act as lipid “carriers” for systemic delivery. Although LNPs can be designed to show some tissue tropism, this is often limited to organs such as the liver, spleen, and lungs.
 Dr. Zak[/caption]
MedicalResearch.com Interview with:
Magdalena M. Żak, PhD
Zangi Lab
Instructor | Cardiovascular Research Institute
Instructor | Genetics & Genomic Sciences
Icahn School of Medicine at Mount Sinai
Hess Center for Science and Medicine
New York, NY 10029
MedicalResearch.com: What is the background for this study?
Response: mRNA has proven to be a groundbreaking technology through COVID-19 vaccines, and most mRNA-based therapeutics in development today are still focused on vaccines. However, in principle, mRNA could be used for many diseases in which expression of a therapeutic protein would be beneficial. A major reason mRNA is less commonly used outside of vaccines is the lack of robust targeting: for vaccination, broad expression can be acceptable because the goal is antigen production for immune recognition, but for other applications - especially cancer - targeted delivery and minimized off-target expression are critical to reduce side effects. Current targeting strategies largely rely on lipid nanoparticles (LNPs), which act as lipid “carriers” for systemic delivery. Although LNPs can be designed to show some tissue tropism, this is often limited to organs such as the liver, spleen, and lungs.
Dr. Zak[/caption]
MedicalResearch.com Interview with:
Magdalena M. Żak, PhD
Zangi Lab
Instructor | Cardiovascular Research Institute
Instructor | Genetics & Genomic Sciences
Icahn School of Medicine at Mount Sinai
Hess Center for Science and Medicine
New York, NY 10029
MedicalResearch.com: What is the background for this study?
Response: mRNA has proven to be a groundbreaking technology through COVID-19 vaccines, and most mRNA-based therapeutics in development today are still focused on vaccines. However, in principle, mRNA could be used for many diseases in which expression of a therapeutic protein would be beneficial. A major reason mRNA is less commonly used outside of vaccines is the lack of robust targeting: for vaccination, broad expression can be acceptable because the goal is antigen production for immune recognition, but for other applications - especially cancer - targeted delivery and minimized off-target expression are critical to reduce side effects. Current targeting strategies largely rely on lipid nanoparticles (LNPs), which act as lipid “carriers” for systemic delivery. Although LNPs can be designed to show some tissue tropism, this is often limited to organs such as the liver, spleen, and lungs.


 Dr. Campanella[/caption]
Gabriele Campanella, PhD
Assistant Professor
Windreich Department of Artificial Intelligence and Human Health
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the most lethal cancer in the US. Lung adenocarcinoma (LUAD) is the most common form of lung cancer with an incidence of over 100k per year in the US. EGFR mutations are common driver mutations in LUAD, and importantly, these mutations can be targeted by TKI therapy, which has high response rates. Because of this, EGFR testing via NGS (Next Generation Sequencing) is considered mandatory by guidelines for any LUAD diagnosis.
In high-resource settings, rapid EGFR testing is done while waiting for confirmation via NGS. This is because NGS takes about 2 weeks on average, while the rapid testing has a median TAT of 2 days. Early treatment decisions could be made based on the rapid test results. Rapid tests have some important drawbacks, most notably, it exhausts tissue. In lung cancer, tissue is scarce in the first place, and up to 25% of cases, after rapid testing there is not enough tissue for NGS. In those circumstances, patients have to be biopsied again, which adds unnecessary risk for the patient. Even worse, in some cases, the NGS is never done. A non-tissue-exhaustive computational biomarker could be used instead of the tissue-based rapid test.
Dr. Campanella[/caption]
Gabriele Campanella, PhD
Assistant Professor
Windreich Department of Artificial Intelligence and Human Health
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the most lethal cancer in the US. Lung adenocarcinoma (LUAD) is the most common form of lung cancer with an incidence of over 100k per year in the US. EGFR mutations are common driver mutations in LUAD, and importantly, these mutations can be targeted by TKI therapy, which has high response rates. Because of this, EGFR testing via NGS (Next Generation Sequencing) is considered mandatory by guidelines for any LUAD diagnosis.
In high-resource settings, rapid EGFR testing is done while waiting for confirmation via NGS. This is because NGS takes about 2 weeks on average, while the rapid testing has a median TAT of 2 days. Early treatment decisions could be made based on the rapid test results. Rapid tests have some important drawbacks, most notably, it exhausts tissue. In lung cancer, tissue is scarce in the first place, and up to 25% of cases, after rapid testing there is not enough tissue for NGS. In those circumstances, patients have to be biopsied again, which adds unnecessary risk for the patient. Even worse, in some cases, the NGS is never done. A non-tissue-exhaustive computational biomarker could be used instead of the tissue-based rapid test.

 Dr. Mzoughi[/caption]
Slim Mzoughi, PhD
Dr. Mzoughi[/caption]
Slim Mzoughi, PhD




 Ben Petrazzini[/caption]
Ben Omega Petrazzini, B.Sc.
Associate Bioinformatician
Ben Petrazzini[/caption]
Ben Omega Petrazzini, B.Sc.
Associate Bioinformatician

 Dr. Patel[/caption]
Rima Patel, MD
Assistant Professor, Division of Hematology/Oncology
The Tisch Cancer Institute
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: The 21-gene Oncotype DX Recurrence Score (RS) and 70-gene MammaPrint (MP) assays provide prognostic information for distant recurrence and are used to guide chemotherapy use in hormone receptor (HR)-positive, HER2-negative early breast cancer (EBC). Previous reports have demonstrated racial differences in the prognostic accuracy of the RS. In both the TAILORx and RxPONDER trials, Black women with low genomic risk (RS 0-25) had a higher recurrence risk than White women. In another study using the NCDB database,
Dr. Patel[/caption]
Rima Patel, MD
Assistant Professor, Division of Hematology/Oncology
The Tisch Cancer Institute
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: The 21-gene Oncotype DX Recurrence Score (RS) and 70-gene MammaPrint (MP) assays provide prognostic information for distant recurrence and are used to guide chemotherapy use in hormone receptor (HR)-positive, HER2-negative early breast cancer (EBC). Previous reports have demonstrated racial differences in the prognostic accuracy of the RS. In both the TAILORx and RxPONDER trials, Black women with low genomic risk (RS 0-25) had a higher recurrence risk than White women. In another study using the NCDB database, 



 Dr. Gaba[/caption]
Prakriti Gaba, MD
Cardiovascular Medicine Fellow
Brigham and Women's Hospital
[caption id="attachment_31114" align="alignleft" width="120"]
Dr. Gaba[/caption]
Prakriti Gaba, MD
Cardiovascular Medicine Fellow
Brigham and Women's Hospital
[caption id="attachment_31114" align="alignleft" width="120"] Dr. Deepak Bhatt[/caption]
Deepak L. Bhatt MD MPH
Director of Mount Sinai Fuster Heart Hospital
Dr. Valentin Fuster Professor of Cardiovascular Medicine
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: Accurate classification of acute myocardial infarction is instrumental for the appropriate diagnosis and effective management of patients suffering from this widely prevalent cardiovascular condition.
In the past, there have been a variety of clinical scores published to advise clinicians on the best classifications schemes for patients with acute MI. These have included the Thrombolysis in Myocardial Infarction (TIMI) risk score, the HEART score, and the Killip classification. The strength of these traditional scores includes their practicality, as they can be implemented at the bedside to rapidly assist with prognostication. Nonetheless, as technologic advancements have made imaging and tissue identification more accessible, national and international committees are looking to revise traditional classification schemes of acute MI with novel ones leveraging multimodal approaches.
Dr. Deepak Bhatt[/caption]
Deepak L. Bhatt MD MPH
Director of Mount Sinai Fuster Heart Hospital
Dr. Valentin Fuster Professor of Cardiovascular Medicine
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: Accurate classification of acute myocardial infarction is instrumental for the appropriate diagnosis and effective management of patients suffering from this widely prevalent cardiovascular condition.
In the past, there have been a variety of clinical scores published to advise clinicians on the best classifications schemes for patients with acute MI. These have included the Thrombolysis in Myocardial Infarction (TIMI) risk score, the HEART score, and the Killip classification. The strength of these traditional scores includes their practicality, as they can be implemented at the bedside to rapidly assist with prognostication. Nonetheless, as technologic advancements have made imaging and tissue identification more accessible, national and international committees are looking to revise traditional classification schemes of acute MI with novel ones leveraging multimodal approaches.

 Dr. Dubinsky[/caption]
Marla C. Dubinsky, MD
Professor of Pediatrics and Medicine
Icahn School of Medicine at Mount Sinai
Co- director, Susan and Leonard Feinstein IBD Clinical Center
Mount Sinai Health System
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of UC?
Response: Lucent 1 and Lucent 2 were the induction and maintenance registration trials studying the efficacy and safety of mirikizumab in patients 18 years and older with moderate to severely active ulcerative colitis. Mirikizumab is a monoclonal antibody targeting the p19 subunit of IL23. Lucent-3 is the open label extension arm for those meeting inclusion criteria after completing Lucent 2. This study evaluated the long term efficacy and safety of mirikizumab in patients with ulcerative colitis who completed a total of 104 weeks of active mirikizumab treatment.
Ulcerative colitis is a chronic incurable inflammatory condition of colon. Common symptoms include diarrhea, blood in the stool, abdominal cramping and bowel urgency. Bowel urgency is one of the most burdensome symptoms that a patient with you could experience.
Dr. Dubinsky[/caption]
Marla C. Dubinsky, MD
Professor of Pediatrics and Medicine
Icahn School of Medicine at Mount Sinai
Co- director, Susan and Leonard Feinstein IBD Clinical Center
Mount Sinai Health System
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of UC?
Response: Lucent 1 and Lucent 2 were the induction and maintenance registration trials studying the efficacy and safety of mirikizumab in patients 18 years and older with moderate to severely active ulcerative colitis. Mirikizumab is a monoclonal antibody targeting the p19 subunit of IL23. Lucent-3 is the open label extension arm for those meeting inclusion criteria after completing Lucent 2. This study evaluated the long term efficacy and safety of mirikizumab in patients with ulcerative colitis who completed a total of 104 weeks of active mirikizumab treatment.
Ulcerative colitis is a chronic incurable inflammatory condition of colon. Common symptoms include diarrhea, blood in the stool, abdominal cramping and bowel urgency. Bowel urgency is one of the most burdensome symptoms that a patient with you could experience.


 Prof. Dubinsky[/caption]
Marla Dubinsky, M.D.
Professor of Pediatrics and Medicine
Co-director of the Susan and Leonard Feinstein IBD Clinical Center
Chief of the Division of Pediatric Gastroenterology and Nutrition
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? How does MIRIKIZUMAB differ from other medications for UC?
Response: This is a phase 2 study to assess the PK (pharamcokinetics), safety and efficacy of mirikizumab in pediatric ulcerative colitis (UC).
Prof. Dubinsky[/caption]
Marla Dubinsky, M.D.
Professor of Pediatrics and Medicine
Co-director of the Susan and Leonard Feinstein IBD Clinical Center
Chief of the Division of Pediatric Gastroenterology and Nutrition
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? How does MIRIKIZUMAB differ from other medications for UC?
Response: This is a phase 2 study to assess the PK (pharamcokinetics), safety and efficacy of mirikizumab in pediatric ulcerative colitis (UC).


 Dr. Guttman-Yassky[/caption]
Dr. Emma Guttman-Yassky, MD, PhD
Waldman Professor and System Chair
The Kimberly and Eric J. Waldman Department of Dermatology
Director, Center of Excellence in Eczema
Director, Laboratory of Inflammatory Skin Diseases
Icahn School of Medicine at Mount Sinai
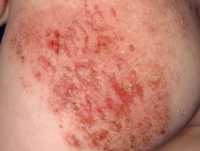
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The background for this study are studies that show that OX40 is a pathway that is upregulated in patients with atopic dermatitis (or eczema). OX40 is involved in activation of immune molecules associated with allergy and atopy, and also with formation of memory immune cells that are required for disease recurrence. The hypothesis to the study was that giving an OX40 antagonist will not only ameliorate the disease but perhaps have a remittive effect in that the disease will not come back.
Indeed all drug doses were significantly effective at week 16, the primary endpoint compared to placebo and continued to improve towards week 36, the secondary endpoint. In addition, the responders to treatment maintained their responses for an additional 20 weeks, which is unusual, suggesting a potential for disease modification.
Dr. Guttman-Yassky[/caption]
Dr. Emma Guttman-Yassky, MD, PhD
Waldman Professor and System Chair
The Kimberly and Eric J. Waldman Department of Dermatology
Director, Center of Excellence in Eczema
Director, Laboratory of Inflammatory Skin Diseases
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The background for this study are studies that show that OX40 is a pathway that is upregulated in patients with atopic dermatitis (or eczema). OX40 is involved in activation of immune molecules associated with allergy and atopy, and also with formation of memory immune cells that are required for disease recurrence. The hypothesis to the study was that giving an OX40 antagonist will not only ameliorate the disease but perhaps have a remittive effect in that the disease will not come back.
Indeed all drug doses were significantly effective at week 16, the primary endpoint compared to placebo and continued to improve towards week 36, the secondary endpoint. In addition, the responders to treatment maintained their responses for an additional 20 weeks, which is unusual, suggesting a potential for disease modification.

 Dr. Midya[/caption]
Dania Valvi, MD MPH PhD
Assistant Professor
Department of Environmental Medicine and Public Health
Co-Director, MS in Epidemiology
Icahn School of Medicine at Mount Sinai
Email: dania.valvi@mssm.edu
MedicalResearch.com: What is the background for this study?
Response: Non-alcoholic fatty liver disease (NAFLD) is the most common liver disease in children in the U.S., Europe and other world regions, currently affecting 1 in every 10 children, and 1 in every 3 children with obesity in the U.S. The rate of pediatric NAFLD has more than doubled in recent decades following the epidemic rates also noted for childhood obesity. There is increasing interest in the role that environmental chemical exposures may play in NAFLD etiology, since several animal studies have shown that prenatal exposures to endocrine-disrupting chemicals (EDCs) cause liver injury and damage; but, until now, the potential effects of prenatal EDC mixture exposures in pediatric NAFLD had not been studied.
Dr. Midya[/caption]
Dania Valvi, MD MPH PhD
Assistant Professor
Department of Environmental Medicine and Public Health
Co-Director, MS in Epidemiology
Icahn School of Medicine at Mount Sinai
Email: dania.valvi@mssm.edu
MedicalResearch.com: What is the background for this study?
Response: Non-alcoholic fatty liver disease (NAFLD) is the most common liver disease in children in the U.S., Europe and other world regions, currently affecting 1 in every 10 children, and 1 in every 3 children with obesity in the U.S. The rate of pediatric NAFLD has more than doubled in recent decades following the epidemic rates also noted for childhood obesity. There is increasing interest in the role that environmental chemical exposures may play in NAFLD etiology, since several animal studies have shown that prenatal exposures to endocrine-disrupting chemicals (EDCs) cause liver injury and damage; but, until now, the potential effects of prenatal EDC mixture exposures in pediatric NAFLD had not been studied.










 Dr. Ritch[/caption]
Robert Ritch, MD, FACS, FARVO
Shelley and Steven Einhorn Distinguished Chair
Professor of Ophthalmology, Surgeon Director Emeritus
Chief, Glaucoma Services Emeritus
The New York Eye and Ear Infirmary of Mount Sinai
New York, NY 10003
Founder, Medical Director and
Chairman, Scientific Advisory Board
The Glaucoma Foundation
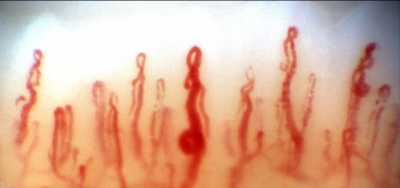
MedicalResearch.com: What is the background for this study?
Response: Nailfold capillaroscopy (NFC), long used in rheumatology is a new approach to investigation of glaucoma.
Posterior to the nailbed and just anterior to the proximal nailfold is the cuticle, which has no structural elements visible to the naked eye. NFC is a non-invasive imaging modality that provides a highly magnified view of the capillaries at the nailfold of digits. It has also been used in ophthalmology to show morphological changes at the nailfold capillaries of POAG and XFG/XFS patients, helping to confirm the systemic nature of these diseases.
[caption id="attachment_52469" align="alignleft" width="400"]
Dr. Ritch[/caption]
Robert Ritch, MD, FACS, FARVO
Shelley and Steven Einhorn Distinguished Chair
Professor of Ophthalmology, Surgeon Director Emeritus
Chief, Glaucoma Services Emeritus
The New York Eye and Ear Infirmary of Mount Sinai
New York, NY 10003
Founder, Medical Director and
Chairman, Scientific Advisory Board
The Glaucoma Foundation
MedicalResearch.com: What is the background for this study?
Response: Nailfold capillaroscopy (NFC), long used in rheumatology is a new approach to investigation of glaucoma.
Posterior to the nailbed and just anterior to the proximal nailfold is the cuticle, which has no structural elements visible to the naked eye. NFC is a non-invasive imaging modality that provides a highly magnified view of the capillaries at the nailfold of digits. It has also been used in ophthalmology to show morphological changes at the nailfold capillaries of POAG and XFG/XFS patients, helping to confirm the systemic nature of these diseases.
[caption id="attachment_52469" align="alignleft" width="400"]