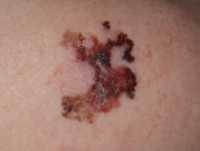
Author Interviews, Cancer Research, ESMO, Race/Ethnic Diversity, Social Issues / 21.09.2020
Minorities At Increased Risk of Cancer Care Disruption During COVID-19
MedicalResearch.com Interview with:
Ziad Bakouny, MD, MSc
Post-doctoral research fellow
Lank Center for Genitourinary Oncology
Dana-Farber Cancer Institute
MedicalResearch.com: What is the background for this study?
Response: The COVID-19 pandemic has disproportionately affected patients with cancer, with these patients unfortunately having worse outcomes than the general population. In fact, a recent report by the COVID-19 and Cancer Consortium (CCC19) showed that the mortality rate in patients with cancer who develop COVID-19, at 30 days median follow-up, was 16%. Although the adverse outcomes of patients with cancer who develop COVID-19 has received much attention, few studies have thus far investigated the effects of the potential disruption to cancer care delivery caused by the pandemic.
Our aim in the COVID and Cancer Outcomes Study (CCOS) was therefore to evaluate this disruption to cancer care caused by the pandemic. This is a multicenter prospective cohort study that included patients seen in the outpatient setting at the Tisch Cancer Institute, Icahn School of Medicine at Mount Sinai in New York City and the Dana-Farber Cancer Institute in Boston during one week in March (between March 2 and March 6 2020). Data was collected 3 months before this index week and 3 months prospectively (during the first peak of the pandemic in the Northeastern United States). (more…)