MedicalResearch.com Interview with:
Dr Dilip Kachhawa, MD
Department of Skin & Venereal Disease
Dr Sampurnanand Medical College
Jodhpur, Rajasthan, India
MedicalResearch.com: What are the main findings?
[caption id="attachment_59328" align="alignleft" width="150"]
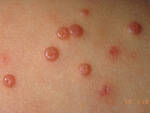
Molluscum DermNet image[/caption]
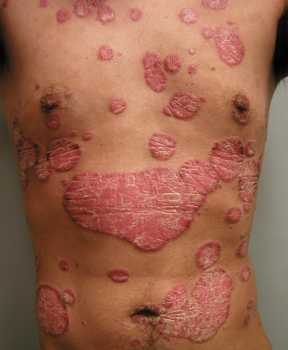
Response: Molluscum Contagiosum (MC) is an infection caused by molluscipoxvirus. It is difficult to study since the virus only survives in human skin, and therefore there isn’t an animal or cell model to study potential treatments. Molluscum lesions appear as raised, domed shaped skin-colored lesions and can occur anywhere on the body but are most common on the face, neck, arms, legs, and abdomen. Sometimes there are few lesions, but clusters of several lesions can appear. Children are the most likely to get molluscum, and the virus is highly contagious, transmitted by direct contact with infected skin or contaminated objects, like towels, linens and toys. Scratching can cause autoinoculation which is when a person reinfects themself.
MC is very common, impacting an estimated 6 million adults and mostly children in the US each year. In 2010, there was an estimated 122 million cases worldwide. It occurs primarily in humid and warm climates, and transmission via swimming pools and bathtubs may be possible. Therefore, molluscum is often called “water warts.”
Many physicians may take a “watch and wait” approach since the virus may clear on its own. However, it can take months to up to 5 years for some to experience complete clearance, In the meantime, the person is still highly contagious and may spread the virus to others, particularly children. Lesions can be bothersome, causing itching and sometimes a secondary infection. There is also a psychosocial component. In a recent study, 1 in 10 children with molluscum experienced a major quality of life issue.
Berdazimer Gel, 10.3% is a potential first-in-class topical controlled-nitric oxide releasing medication containing Berdazimer (sodium), a new chemical entity, and the active ingredient in berdazimer gel 10.3%. The mechanism of action of berdazimer in the treatment of molluscum is unknown, but in vitro lab studies show that the nitric oxide, released when berdazimer is combined with a hydrogel, may impede viral replication and perhaps help body’s natural immune response against molluscum.


 Dr. Guttman-Yassky[/caption]
Dr. Emma Guttman-Yassky, MD, PhD
Waldman Professor and System Chair
The Kimberly and Eric J. Waldman Department of Dermatology
Director, Center of Excellence in Eczema
Director, Laboratory of Inflammatory Skin Diseases
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
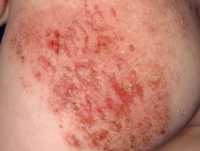
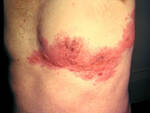
Response: The background for this study are studies that show that OX40 is a pathway that is upregulated in patients with atopic dermatitis (or eczema). OX40 is involved in activation of immune molecules associated with allergy and atopy, and also with formation of memory immune cells that are required for disease recurrence. The hypothesis to the study was that giving an OX40 antagonist will not only ameliorate the disease but perhaps have a remittive effect in that the disease will not come back.
Indeed all drug doses were significantly effective at week 16, the primary endpoint compared to placebo and continued to improve towards week 36, the secondary endpoint. In addition, the responders to treatment maintained their responses for an additional 20 weeks, which is unusual, suggesting a potential for disease modification.
Dr. Guttman-Yassky[/caption]
Dr. Emma Guttman-Yassky, MD, PhD
Waldman Professor and System Chair
The Kimberly and Eric J. Waldman Department of Dermatology
Director, Center of Excellence in Eczema
Director, Laboratory of Inflammatory Skin Diseases
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The background for this study are studies that show that OX40 is a pathway that is upregulated in patients with atopic dermatitis (or eczema). OX40 is involved in activation of immune molecules associated with allergy and atopy, and also with formation of memory immune cells that are required for disease recurrence. The hypothesis to the study was that giving an OX40 antagonist will not only ameliorate the disease but perhaps have a remittive effect in that the disease will not come back.
Indeed all drug doses were significantly effective at week 16, the primary endpoint compared to placebo and continued to improve towards week 36, the secondary endpoint. In addition, the responders to treatment maintained their responses for an additional 20 weeks, which is unusual, suggesting a potential for disease modification.
 Dr. Curhan[/caption]
Sharon G. Curhan, MD, ScM|
Director, CHEARS: The Conservation of Hearing Study
Channing Division of Network Medicine
Department of Medicine
Brigham and Women’s Hospital
Harvard Medical School
Boston, MA 02114
MedicalResearch.com: What is the background for this study?
Response: Herpes zoster, commonly known as “shingles,” is a viral infection that often causes a painful rash. Shingles can occur anywhere on the head or body. Shingles is caused by the varicella zoster virus (VZV), the same virus that causes chickenpox. After a person has chickenpox, the virus stays in their body for the rest of their life. Years and even decades later, the virus may reactivate as shingles. Almost all individuals age 50 years and older in the US have been infected with the varicella zoster virus and therefore they are at risk for shingles.
About 1 in 3 people will develop shingles during their lifetime, and since age is a risk factor for shingles, this number may increase as the population ages. The risk is also higher among individuals of any age who are immunocompromised due to disease or treatment. A number of serious complications can occur when a person develops shingles, such as post-herpetic neuralgia (long-lasting pain), but there was limited information on whether there are other adverse long-term health implications of developing shingles.
There is a growing body of evidence that links VZV, the virus that causes shingles, to vascular disease. VZV vasculopathy may cause damage to blood vessels and increase the risk of stroke or coronary heart disease. Although some previous studies showed a higher risk of stroke or heart attack around the time of the shingles infection, it was not known whether this higher risk persisted in the long term. Therefore, the question we aimed to address in this study was to investigate whether shingles is associated with higher long-term risk of stroke or coronary heart disease.
To address this question, we conducted a prospective longitudinal study in 3 large US cohorts of >200,000 women and men, the Nurses’ Health Study (>79,000 women), the Nurses’ Health Study II (almost 94,000 women) and the Health Professionals Follow-Up Study (>31,000 men), without a prior history of stroke or coronary heart disease. We collected information on shingles, stroke and coronary heart disease on biennial questionnaires and confirmed the diagnoses with medical record review. We followed the participants for up to 16 years and evaluated whether those who had developed shingles were at higher risk for stroke or coronary heart disease years after the shingles episode. The outcomes we measured were incident stroke, incident coronary heart disease [defined as having a non-fatal or fatal myocardial infarction (heart attack) or a coronary revascularization procedure (CABG, coronary artery bypass graft or percutaneous transluminal coronary angioplasty)]. We also evaluated a combined outcome of cardiovascular disease, which included either stroke or coronary heart disease, whichever came first.
Dr. Curhan[/caption]
Sharon G. Curhan, MD, ScM|
Director, CHEARS: The Conservation of Hearing Study
Channing Division of Network Medicine
Department of Medicine
Brigham and Women’s Hospital
Harvard Medical School
Boston, MA 02114
MedicalResearch.com: What is the background for this study?
Response: Herpes zoster, commonly known as “shingles,” is a viral infection that often causes a painful rash. Shingles can occur anywhere on the head or body. Shingles is caused by the varicella zoster virus (VZV), the same virus that causes chickenpox. After a person has chickenpox, the virus stays in their body for the rest of their life. Years and even decades later, the virus may reactivate as shingles. Almost all individuals age 50 years and older in the US have been infected with the varicella zoster virus and therefore they are at risk for shingles.
About 1 in 3 people will develop shingles during their lifetime, and since age is a risk factor for shingles, this number may increase as the population ages. The risk is also higher among individuals of any age who are immunocompromised due to disease or treatment. A number of serious complications can occur when a person develops shingles, such as post-herpetic neuralgia (long-lasting pain), but there was limited information on whether there are other adverse long-term health implications of developing shingles.
There is a growing body of evidence that links VZV, the virus that causes shingles, to vascular disease. VZV vasculopathy may cause damage to blood vessels and increase the risk of stroke or coronary heart disease. Although some previous studies showed a higher risk of stroke or heart attack around the time of the shingles infection, it was not known whether this higher risk persisted in the long term. Therefore, the question we aimed to address in this study was to investigate whether shingles is associated with higher long-term risk of stroke or coronary heart disease.
To address this question, we conducted a prospective longitudinal study in 3 large US cohorts of >200,000 women and men, the Nurses’ Health Study (>79,000 women), the Nurses’ Health Study II (almost 94,000 women) and the Health Professionals Follow-Up Study (>31,000 men), without a prior history of stroke or coronary heart disease. We collected information on shingles, stroke and coronary heart disease on biennial questionnaires and confirmed the diagnoses with medical record review. We followed the participants for up to 16 years and evaluated whether those who had developed shingles were at higher risk for stroke or coronary heart disease years after the shingles episode. The outcomes we measured were incident stroke, incident coronary heart disease [defined as having a non-fatal or fatal myocardial infarction (heart attack) or a coronary revascularization procedure (CABG, coronary artery bypass graft or percutaneous transluminal coronary angioplasty)]. We also evaluated a combined outcome of cardiovascular disease, which included either stroke or coronary heart disease, whichever came first.

 Dr. Batres[/caption]
Carlota Batres, Ph.D.
Dr. Batres[/caption]
Carlota Batres, Ph.D.

 Dr. White[/caption]
Alexandra J. White, PhD, MSPH
Stadtman Investigator
Epidemiology Branch
National Institute of Environmental Health Sciences
National Institutes of Health
Research Triangle Park, NC
MedicalResearch.com: What is the background for this study? What is the chemical primarily used in hair straighteners?
Response: Hair products such as dye and chemical straighteners contain several different chemicals that may act as carcinogens or endocrine disruptors and thus may be important for cancer risk. Straighteners in particular have been found to include chemicals such as phthalates, parabens, cyclosiloxanes and metals and may release formaldehyde when heated. Previous research has suggested that hair dye and chemical straighteners are related to other hormone-sensitive cancers such as breast and ovarian cancer, but no previous study has considered how they are related to uterine cancer risk.
Dr. White[/caption]
Alexandra J. White, PhD, MSPH
Stadtman Investigator
Epidemiology Branch
National Institute of Environmental Health Sciences
National Institutes of Health
Research Triangle Park, NC
MedicalResearch.com: What is the background for this study? What is the chemical primarily used in hair straighteners?
Response: Hair products such as dye and chemical straighteners contain several different chemicals that may act as carcinogens or endocrine disruptors and thus may be important for cancer risk. Straighteners in particular have been found to include chemicals such as phthalates, parabens, cyclosiloxanes and metals and may release formaldehyde when heated. Previous research has suggested that hair dye and chemical straighteners are related to other hormone-sensitive cancers such as breast and ovarian cancer, but no previous study has considered how they are related to uterine cancer risk.




 Dr. Vashi[/caption]
Dr. Neelam Vashi MD
Director of the Boston University Center for Ethnic Skin
Dermatologist at Boston Medical Center, and
[caption id="attachment_59079" align="alignleft" width="100"]
Dr. Vashi[/caption]
Dr. Neelam Vashi MD
Director of the Boston University Center for Ethnic Skin
Dermatologist at Boston Medical Center, and
[caption id="attachment_59079" align="alignleft" width="100"] Dr. De La Garza[/caption]
Dr. Henriette De La Garza MD
Research fellow
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: The COVID-19 pandemic abruptly shifted many of our daily activities to an online world, dramatically increasing the use of electronic devices. Although visible light exposure from screens is small compared with the amount of exposure from the sun, there is concern about the long-term effects of excessive screen time. Recent studies have demonstrated that exposure to light emitted from electronic devices, even for as little as 1 hour, may cause reactive oxygen species generation, apoptosis, collagen degradation, and necrosis of skin cells. Visible light increases tyrosinase activity and induces immediate erythema in light-skinned individuals and long-lasting pigmentation in dark-skinned individuals. In recent years, tinted sunscreens have been rising in popularity because they are an effective and convenient way to protect against high-energy visible light while providing cosmetic benefits. The purpose of this analysis was to study current available options and product factors that may influence consumer preference when choosing a tinted sunscreen so dermatologists can improve their familiarity with available products and tailor their recommendations to patients with all skin tones.
Dr. De La Garza[/caption]
Dr. Henriette De La Garza MD
Research fellow
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: The COVID-19 pandemic abruptly shifted many of our daily activities to an online world, dramatically increasing the use of electronic devices. Although visible light exposure from screens is small compared with the amount of exposure from the sun, there is concern about the long-term effects of excessive screen time. Recent studies have demonstrated that exposure to light emitted from electronic devices, even for as little as 1 hour, may cause reactive oxygen species generation, apoptosis, collagen degradation, and necrosis of skin cells. Visible light increases tyrosinase activity and induces immediate erythema in light-skinned individuals and long-lasting pigmentation in dark-skinned individuals. In recent years, tinted sunscreens have been rising in popularity because they are an effective and convenient way to protect against high-energy visible light while providing cosmetic benefits. The purpose of this analysis was to study current available options and product factors that may influence consumer preference when choosing a tinted sunscreen so dermatologists can improve their familiarity with available products and tailor their recommendations to patients with all skin tones.
 Dr. Ferris[/caption]
Laura Ferris, M.D., Ph.D.
Professor of Dermatology
Director of clinical trials for UPMC Department of Dermatology
University of Pittsburgh School of Medicine
MedicalResearch.com: What is the background for this study?
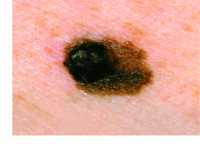
Response: In this quality initiative at UPMC (a large academic and community health system in Western PA and surrounding areas) Primary Care Physicians were trained to perform annual skin cancer screening examinations of their patients who were aged 35 years and older at routine medical visits.
Over a 5-year period more than 595,000 patients who were eligible to be screened were seen by a UPMC PCP and about 24% of them were screened. We compared the number and thickness (an important indicator of prognosis) of the melanomas diagnosed in those patients who were screened to those who were not screened.
Dr. Ferris[/caption]
Laura Ferris, M.D., Ph.D.
Professor of Dermatology
Director of clinical trials for UPMC Department of Dermatology
University of Pittsburgh School of Medicine
MedicalResearch.com: What is the background for this study?
Response: In this quality initiative at UPMC (a large academic and community health system in Western PA and surrounding areas) Primary Care Physicians were trained to perform annual skin cancer screening examinations of their patients who were aged 35 years and older at routine medical visits.
Over a 5-year period more than 595,000 patients who were eligible to be screened were seen by a UPMC PCP and about 24% of them were screened. We compared the number and thickness (an important indicator of prognosis) of the melanomas diagnosed in those patients who were screened to those who were not screened.
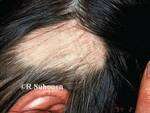
 Brett King, MD, PhD, FAAD
Associate Professor of Dermatology
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
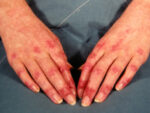
Response: Alopecia areata is an autoimmune disorder marked by disfiguring, non-scarring hair loss, and there are no therapies approved by the U.S. Food and Drug Administration for treatment of the disease. JAK inhibitors are showing promise for treatment of severe alopecia areata. In this work, the pooled results of two phase 3 clinical trials of the JAK inhibitor baricitinib were reported out to 52 weeks.
Brett King, MD, PhD, FAAD
Associate Professor of Dermatology
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Alopecia areata is an autoimmune disorder marked by disfiguring, non-scarring hair loss, and there are no therapies approved by the U.S. Food and Drug Administration for treatment of the disease. JAK inhibitors are showing promise for treatment of severe alopecia areata. In this work, the pooled results of two phase 3 clinical trials of the JAK inhibitor baricitinib were reported out to 52 weeks.


 Dr. Chen[/caption]
Chih-Shan Jason Chen, MD, PhD
Director, Dermatologic and Mohs Micrographic Surgery Unit
Memorial Sloan Kettering Skin Cancer Center at Hauppauge
Attending Mohs Surgeon, Dermatology Service
Memorial Sloan Kettering Cancer Center
Chief, Dermatologic Surgery Northport VA Medical Center
Associate Professor of Clinical Dermatology
Renaissance School of Medicine at Stony Brook University
MedicalResearch.com: What is the background for this study?
Response: Managing a surgical wound on the lower leg can be a challenge. Often, higher wound tension, atrophic skin, edema, and compromised circulation result in higher risks of wound dehiscence and infection, and significantly limit the capacity of wound closure post-surgically. Therefore, healing by secondary intention is a practical option for many lower leg Mohs defects. However, a secondary intention wound on the lower leg is expected to take a longer time to heal. Certain factors such as older age and health conditions of the host may adversely affect healing time.
Timolol is a nonselective beta-adrenergic receptor antagonist that has FDA approval for the treatment of glaucoma. In addition to this FDA-approved indication, topical timolol has several off-label uses in dermatology, such as for the treatment of infantile hemangiomas, venous stasis ulcers, and refractory wounds. Although timolol solution has been used in chronic wounds, knowledges of the efficacy and utility of timolol in an acute post-surgical wound setting is lacking.
Dr. Chen[/caption]
Chih-Shan Jason Chen, MD, PhD
Director, Dermatologic and Mohs Micrographic Surgery Unit
Memorial Sloan Kettering Skin Cancer Center at Hauppauge
Attending Mohs Surgeon, Dermatology Service
Memorial Sloan Kettering Cancer Center
Chief, Dermatologic Surgery Northport VA Medical Center
Associate Professor of Clinical Dermatology
Renaissance School of Medicine at Stony Brook University
MedicalResearch.com: What is the background for this study?
Response: Managing a surgical wound on the lower leg can be a challenge. Often, higher wound tension, atrophic skin, edema, and compromised circulation result in higher risks of wound dehiscence and infection, and significantly limit the capacity of wound closure post-surgically. Therefore, healing by secondary intention is a practical option for many lower leg Mohs defects. However, a secondary intention wound on the lower leg is expected to take a longer time to heal. Certain factors such as older age and health conditions of the host may adversely affect healing time.
Timolol is a nonselective beta-adrenergic receptor antagonist that has FDA approval for the treatment of glaucoma. In addition to this FDA-approved indication, topical timolol has several off-label uses in dermatology, such as for the treatment of infantile hemangiomas, venous stasis ulcers, and refractory wounds. Although timolol solution has been used in chronic wounds, knowledges of the efficacy and utility of timolol in an acute post-surgical wound setting is lacking.



 Dr. Woodruff[/caption]
Carina M. Woodruff, MD
Department of Dermatolog
University of California, San Francisco
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Rigorous hand hygiene has been an important component of the CDC's COVID-19 guidelines. With millions of Americans now using hand sanitizers regularly, we are seeing many more cases of hand dermatitis. Our study evaluated the key product features and most common allergens in the top-reviewed, commercial hand sanitizers sold by major US retailers.
We found that the most common potential allergens were tocopherol, fragrance, propylene glycol and phenoxyethanol. Our study also showed that nearly 1 in 5 marketing claims on these products was misleading. For example, 70% of sanitizers with the marketing claim "hypoallergenic" included at least one common allergen in its formulation.
Dr. Woodruff[/caption]
Carina M. Woodruff, MD
Department of Dermatolog
University of California, San Francisco
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Rigorous hand hygiene has been an important component of the CDC's COVID-19 guidelines. With millions of Americans now using hand sanitizers regularly, we are seeing many more cases of hand dermatitis. Our study evaluated the key product features and most common allergens in the top-reviewed, commercial hand sanitizers sold by major US retailers.
We found that the most common potential allergens were tocopherol, fragrance, propylene glycol and phenoxyethanol. Our study also showed that nearly 1 in 5 marketing claims on these products was misleading. For example, 70% of sanitizers with the marketing claim "hypoallergenic" included at least one common allergen in its formulation.

 Lara van der Schoot[/caption]
Lara van der Schoot
MD, PhD candidate
Department of Dermatology
Radboud University Medical Center
Nijmegen, The Netherlands
MedicalResearch.com: What is the background for this study?
Response: Psoriasis is a chronic, immune mediated skin disease for which effective targeted biological agents have become available the past years. Inherent to their immunomodulatory mechanism of action, biologics might increase infections risk. We know from clinical trial data that respiratory tract infections are among the most common adverse events during biologic treatment, but real-world data is sparse. Regarding the risk of serious infections among biologic users, mostly defined as infections requiring hospitalization, previous studies provided different results and there is limited comparative data for the newer biologics available.
The COVID-19 pandemic turned attention to the risk of infections among biologic users, especially for respiratory tract infections, as they might relate to susceptibility for viral respiratory tract infections such as COVID-19.
In our study, the primary aim was to determine the risk of respiratory tract infections among real-world psoriasis patients treated with biologics, including the newer IL-17 and IL-23 inhibitors. The secondary aim was to assess risk of serious infections in this cohort. Additionally, rates of SARS-CoV-2 infections were assessed.
Lara van der Schoot[/caption]
Lara van der Schoot
MD, PhD candidate
Department of Dermatology
Radboud University Medical Center
Nijmegen, The Netherlands
MedicalResearch.com: What is the background for this study?
Response: Psoriasis is a chronic, immune mediated skin disease for which effective targeted biological agents have become available the past years. Inherent to their immunomodulatory mechanism of action, biologics might increase infections risk. We know from clinical trial data that respiratory tract infections are among the most common adverse events during biologic treatment, but real-world data is sparse. Regarding the risk of serious infections among biologic users, mostly defined as infections requiring hospitalization, previous studies provided different results and there is limited comparative data for the newer biologics available.
The COVID-19 pandemic turned attention to the risk of infections among biologic users, especially for respiratory tract infections, as they might relate to susceptibility for viral respiratory tract infections such as COVID-19.
In our study, the primary aim was to determine the risk of respiratory tract infections among real-world psoriasis patients treated with biologics, including the newer IL-17 and IL-23 inhibitors. The secondary aim was to assess risk of serious infections in this cohort. Additionally, rates of SARS-CoV-2 infections were assessed.

 Dr. Ezhkova[/caption]
Elena Ezhkova, PhD
Professor, Department of Cell, Developmental, and Regenerative Biology
Professsor, Dermatology
Lab Head,The Black Family Stem Cell Institute
Icahn School of Medicine at Mount Sinai
New York
[caption id="attachment_58115" align="alignleft" width="125"]
Dr. Ezhkova[/caption]
Elena Ezhkova, PhD
Professor, Department of Cell, Developmental, and Regenerative Biology
Professsor, Dermatology
Lab Head,The Black Family Stem Cell Institute
Icahn School of Medicine at Mount Sinai
New York
[caption id="attachment_58115" align="alignleft" width="125"] Dr. Li[/caption]
Meng-Yen Li, PhD
Postdoctoral Fellow
The Black Family Stem Cell Institute
MedicalResearch.com: What is the background for this study?
Response: The epidermis is the primary barrier and the first line of defense to combat environmental stressors. The sun's ultraviolet (UV) is one of the main environmental stressors that our body is exposed to daily. UV produces DNA damage in epidermal cells and is a leading cause of skin cancers.
To protect from the damaging effects of UV, epidermal cells become pigmented by melanocytes, pigment-producing cells. Taken up by epidermal cells, the melanin pigment absorbs UV light and reduces DNA damage. How the epidermis senses UV and how it leads to epidermal pigmentation is poorly understood.
Dr. Li[/caption]
Meng-Yen Li, PhD
Postdoctoral Fellow
The Black Family Stem Cell Institute
MedicalResearch.com: What is the background for this study?
Response: The epidermis is the primary barrier and the first line of defense to combat environmental stressors. The sun's ultraviolet (UV) is one of the main environmental stressors that our body is exposed to daily. UV produces DNA damage in epidermal cells and is a leading cause of skin cancers.
To protect from the damaging effects of UV, epidermal cells become pigmented by melanocytes, pigment-producing cells. Taken up by epidermal cells, the melanin pigment absorbs UV light and reduces DNA damage. How the epidermis senses UV and how it leads to epidermal pigmentation is poorly understood.

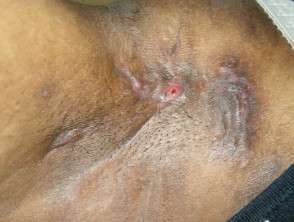
 Dr. Albrecht[/caption]
Joerg Albrecht, MD, PhD
Dermatologist, Internist, Clinical Pharmacologist
Division of Dermatology, Department of Medicine
Attending Dermatologist, Chair Division of Dermatology
Chair system-wide Pharmacy & Therapeutics Committee
Cook County Health
Chicago, IL 60612
MedicalResearch.com: What is the background for this study?
Response: The study was stimulated by data that suggested that an unusually large proportion of inpatient with Hidradenitis suppurativa (HS) were African American. HS is an underdiagnosed disease and the total numbers of inpatients with HS in the year we looked at initially seemed small. So we wanted to test whether this finding held up when the period of observation was extended. Point estimates can be unreliable and we had followed another finding in the data that did not hold up when we looked at other years, so we felt one year was not enough to confirm a trend
Dr. Albrecht[/caption]
Joerg Albrecht, MD, PhD
Dermatologist, Internist, Clinical Pharmacologist
Division of Dermatology, Department of Medicine
Attending Dermatologist, Chair Division of Dermatology
Chair system-wide Pharmacy & Therapeutics Committee
Cook County Health
Chicago, IL 60612
MedicalResearch.com: What is the background for this study?
Response: The study was stimulated by data that suggested that an unusually large proportion of inpatient with Hidradenitis suppurativa (HS) were African American. HS is an underdiagnosed disease and the total numbers of inpatients with HS in the year we looked at initially seemed small. So we wanted to test whether this finding held up when the period of observation was extended. Point estimates can be unreliable and we had followed another finding in the data that did not hold up when we looked at other years, so we felt one year was not enough to confirm a trend

