Aging, Author Interviews, Dermatology / 02.10.2020
Scientists Identify Factor That May Allow Skin to Heal Without Scarring
MedicalResearch.com Interview with:
[caption id="attachment_55555" align="alignleft" width="133"] Dr. Driskell[/caption]
Ryan R. Driskell Ph.D.
Assistant Professor
School of Molecular Biosciences
Center for Reproductive Biology
Washington State University
Pullman, WA. 99164
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Scars a serious health concern when they cover large areas of the body. Understanding how to functionally regenerate skin can improve the quality of life for individuals with large scars.
Importantly, our study is proof of concept, in that we identified a genetic factor that allows adult skin to repair itself like the skin of a newborn babe. The discovery has implications for better skin wound treatment as well as preventing some of the aging process in skin.
Dr. Driskell[/caption]
Ryan R. Driskell Ph.D.
Assistant Professor
School of Molecular Biosciences
Center for Reproductive Biology
Washington State University
Pullman, WA. 99164
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Scars a serious health concern when they cover large areas of the body. Understanding how to functionally regenerate skin can improve the quality of life for individuals with large scars.
Importantly, our study is proof of concept, in that we identified a genetic factor that allows adult skin to repair itself like the skin of a newborn babe. The discovery has implications for better skin wound treatment as well as preventing some of the aging process in skin.
 Dr. Driskell[/caption]
Ryan R. Driskell Ph.D.
Assistant Professor
School of Molecular Biosciences
Center for Reproductive Biology
Washington State University
Pullman, WA. 99164
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Scars a serious health concern when they cover large areas of the body. Understanding how to functionally regenerate skin can improve the quality of life for individuals with large scars.
Importantly, our study is proof of concept, in that we identified a genetic factor that allows adult skin to repair itself like the skin of a newborn babe. The discovery has implications for better skin wound treatment as well as preventing some of the aging process in skin.
Dr. Driskell[/caption]
Ryan R. Driskell Ph.D.
Assistant Professor
School of Molecular Biosciences
Center for Reproductive Biology
Washington State University
Pullman, WA. 99164
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Scars a serious health concern when they cover large areas of the body. Understanding how to functionally regenerate skin can improve the quality of life for individuals with large scars.
Importantly, our study is proof of concept, in that we identified a genetic factor that allows adult skin to repair itself like the skin of a newborn babe. The discovery has implications for better skin wound treatment as well as preventing some of the aging process in skin.




 Dr. Lara-Corrales[/caption]
Irene Lara-Corrales, MD
Associate Professor of Pediatrics at the University of Toronto
Staff physician in Pediatric Dermatology at the
Hospital for Sick Children in Toronto, Canada
She is a member of the
Dr. Lara-Corrales[/caption]
Irene Lara-Corrales, MD
Associate Professor of Pediatrics at the University of Toronto
Staff physician in Pediatric Dermatology at the
Hospital for Sick Children in Toronto, Canada
She is a member of the 
 Dr. Huang[/caption]
Jennifer Huang, MD
Dr. Huang is a pediatric dermatologist at Boston Children’s Hospital and Dana-Farber Cancer Institute.
She is an Associate Professor of Dermatology at Harvard Medical School.
Dr. Huang is a member of the
Dr. Huang[/caption]
Jennifer Huang, MD
Dr. Huang is a pediatric dermatologist at Boston Children’s Hospital and Dana-Farber Cancer Institute.
She is an Associate Professor of Dermatology at Harvard Medical School.
Dr. Huang is a member of the 




 Alyssa M. Thompson[/caption]
Alyssa M. Thompson is currently a 2nd year medical student at the UA-COM Tucson. She graduated from the University of Arizona, Summa Cum Laude in 2018 as the athletic department's Valedictorian with a degree in Physiology and an Entrepreneurship certificate. Her passion for research and dermatology stems from her innovative and integrative mindset with specific interest in inflammatory skin disease.
MedicalResearch.com: What is the background for this study?
Response: Eczema is very common in children. Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products. Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug-store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.
Alyssa M. Thompson[/caption]
Alyssa M. Thompson is currently a 2nd year medical student at the UA-COM Tucson. She graduated from the University of Arizona, Summa Cum Laude in 2018 as the athletic department's Valedictorian with a degree in Physiology and an Entrepreneurship certificate. Her passion for research and dermatology stems from her innovative and integrative mindset with specific interest in inflammatory skin disease.
MedicalResearch.com: What is the background for this study?
Response: Eczema is very common in children. Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products. Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug-store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.

 Dr. Schoch[/caption]
Jennifer Schoch, MD
Dr. Schoch is a pediatric dermatologist and
Associate Professor of Dermatology at the University of Florida.
Her research focuses on the infantile skin microbiome and its role in pediatric skin disease.
She is a member of the Society for Pediatric Dermatology.
[caption id="attachment_54942" align="alignleft" width="144"]
Dr. Schoch[/caption]
Jennifer Schoch, MD
Dr. Schoch is a pediatric dermatologist and
Associate Professor of Dermatology at the University of Florida.
Her research focuses on the infantile skin microbiome and its role in pediatric skin disease.
She is a member of the Society for Pediatric Dermatology.
[caption id="attachment_54942" align="alignleft" width="144"]
 Dr. Harper[/caption]
Julie C. Harper, MD
Clinical Associate Professor of Dermatology
University of Alabama-Birmingham
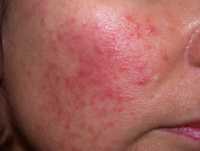
MedicalResearch.com: What is the background for this study?
How common is rosacea?
What are the clinical manifestations of facial rosacea or psoriasis?
Dr. Harper[/caption]
Julie C. Harper, MD
Clinical Associate Professor of Dermatology
University of Alabama-Birmingham
MedicalResearch.com: What is the background for this study?
How common is rosacea?
What are the clinical manifestations of facial rosacea or psoriasis?
![MedicalResearch.com Interview with: Dr Herman Anne Service de Dermatologie Cliniques Universitaires Saint-Luc Avenue Hippocrate, 10 1200 Bruxelle MedicalResearch.com: What is the background for this study? Response: In the context of the COVID-19 pandemic, several cases of acro-located lesions (on foot or hands) suggestive of chilblains have been reported and were possibly related to COVID-19. We wanted to determine if chilblains, observed in many patients recently referred to our department, are indicative of COVID-19. MedicalResearch.com: Would you briefly explain what is meant by chilblains? Response: Chilblains are frequent cold induced inflammatory lesions. Chilblains are typically seen in winter and occur after repeated exposure to cold temperatures. Clinical presentation includes erythema and swelling on toes and/or digits followed by red-purple macules or patches. However, given the large number of patients affected, and the exceptionally high outdoor temperatures for the spring season over the past month and at the time of case-observation, cold-exposure seemed unlikely. These lesions were, therefore, suspected to be associated with COVID-19. However, to date, no study has proven a pathological link between these lesions and COVID-19. MedicalResearch.com: What are the main findings? Response: In our series of 31 patients who recently developed chilblains, negative nasopharyngeal swabs and the absence of anti-SARS-CoV-2 blood immunoglobulin (Ig)M and IgG antibodies in all patients included in your study suggested that these patients had not been infected with COVID-19. These lesions appeared not to be directly related to COVID-19. MedicalResearch.com: What should readers take away from your report? Response: One hypothesis points to an indirect consequence of the COVID-19 pandemic due to imposed community containment and lockdown measures. Indeed, all patients in this study had either been working from home or were home-schooled since the beginning of containment measures in Belgium (March 11, 2020) or were temporarily unemployed. As a result of containment measures, the majority (64.5%) of patients reported a decrease in their physical activity and significantly more time spent in sedentary positions in front of screens. Most of the patients declared that they remained barefoot or in socks most of the day. All these lifestyle changes can be considered as risk factors for developing chilblains. Therefore, it seems plausible that containment, through its collateral effects, may induce chilblains. Interestingly also, the mean BMI of the patients included was relatively low, suggesting that thin people may be more at risk of developing chilblains. MedicalResearch.com: What recommendations do you have for future research as a result of this work? Response: During the current pandemic, several reports have suggested a possible link between cutaneous manifestations including aural lesions such as chilblains, and COVID-19, however, only few patients were tested for SARS-CoV-2 by RT-PCR and no serologic tests were performed. Therefore, reliable testings (RT-PCR and serologic testing) are essential to confirm a potential association with COVID-19. Citation: Herman A, Peeters C, Verroken A, et al. Evaluation of Chilblains as a Manifestation of the COVID-19 Pandemic. JAMA Dermatol. Published online June 25, 2020. doi:10.1001/jamadermatol.2020.2368 https://jamanetwork.com/journals/jamadermatology/fullarticle/2767774 [subscribe] Last Modified: [last-modified] The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2020/06/chilblain-example-dermnet-nz.jpg)


 Dr. Shumel[/caption]
Brad Shumel, MD
Senior Director of Medical Affairs, Immunology
Regeneron
MedicalResearch.com: What is the background for this study?
Response: Atopic dermatitis is a chronic inflammatory disease and one of the most common skin disorders in children. Severe atopic dermatitis is characterized by skin lesions that often cover a large body surface area and can include intense, persistent itch. Uncontrolled moderate-to-severe atopic dermatitis can have a physical, emotional and psychosocial impact on children, resulting in sleep deprivation, activity restriction, poor school performance, depression and anxiety that can have a greater impact on quality-of-life.
The standard of care for this pediatric population has been topical corticosteroids. Children with severe atopic dermatitis who remain uncontrolled with topical therapies have limited treatment options.
This Phase 3 trial was conducted to evaluate the safety and efficacy of dupilumab plus topical corticosteroids (TCS) compared with TCS alone in children with uncontrolled severe atopic dermatitis across two treatment arms – every four weeks and every two weeks (Q4W and Q2W).
Dr. Shumel[/caption]
Brad Shumel, MD
Senior Director of Medical Affairs, Immunology
Regeneron
MedicalResearch.com: What is the background for this study?
Response: Atopic dermatitis is a chronic inflammatory disease and one of the most common skin disorders in children. Severe atopic dermatitis is characterized by skin lesions that often cover a large body surface area and can include intense, persistent itch. Uncontrolled moderate-to-severe atopic dermatitis can have a physical, emotional and psychosocial impact on children, resulting in sleep deprivation, activity restriction, poor school performance, depression and anxiety that can have a greater impact on quality-of-life.
The standard of care for this pediatric population has been topical corticosteroids. Children with severe atopic dermatitis who remain uncontrolled with topical therapies have limited treatment options.
This Phase 3 trial was conducted to evaluate the safety and efficacy of dupilumab plus topical corticosteroids (TCS) compared with TCS alone in children with uncontrolled severe atopic dermatitis across two treatment arms – every four weeks and every two weeks (Q4W and Q2W).

 Dr. Yosipovitch[/caption]
Gil Yosipovitch, MD, Professor
Miami Itch Center
Lennar Medical Foundation
South Miami Clinic in Coral Gables
University of Miami Health System
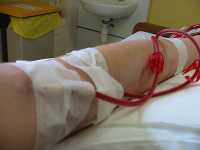
MedicalResearch.com: What is the background for this study?
Response: Chronic Pruritus is a common and burdensome condition in patients with end stage chronic kidney disease (CKD). It is Present at all stages of CKD, not only in patients undergoing hemodialysis (including stage 3-5 CKD). There are no approved treatments for this condition in US and Europe. CKD pruritus has significant impact on quality of life of patients with higher mortality rates due to its effect on sleep.
Studies in the last 2 decades have shown that in patients with CKD pruritus there is an imbalance between endogenous mu opioids that are over expressed to Kappa Opioids that are down regulated.
Difelikefalin (DFK) is a novel peripherally selective kappa opioid receptor (KOR) agonist. Study of IV DFK administration in hemodialysis patients has recently been published and showed significant anti Pruritic effect ( NEJM Fishbane et al. 382: 289-290, 2020).
Dr. Yosipovitch[/caption]
Gil Yosipovitch, MD, Professor
Miami Itch Center
Lennar Medical Foundation
South Miami Clinic in Coral Gables
University of Miami Health System
MedicalResearch.com: What is the background for this study?
Response: Chronic Pruritus is a common and burdensome condition in patients with end stage chronic kidney disease (CKD). It is Present at all stages of CKD, not only in patients undergoing hemodialysis (including stage 3-5 CKD). There are no approved treatments for this condition in US and Europe. CKD pruritus has significant impact on quality of life of patients with higher mortality rates due to its effect on sleep.
Studies in the last 2 decades have shown that in patients with CKD pruritus there is an imbalance between endogenous mu opioids that are over expressed to Kappa Opioids that are down regulated.
Difelikefalin (DFK) is a novel peripherally selective kappa opioid receptor (KOR) agonist. Study of IV DFK administration in hemodialysis patients has recently been published and showed significant anti Pruritic effect ( NEJM Fishbane et al. 382: 289-290, 2020).

 Dr. Paller[/caption]
Amy S Paller, MD
Chair, Department of Dermatology
Director, Skin Biology and Diseases Resource-Based Center
Walter J. Hamlin Professor of Dermatology
Professor of Dermatology and Pediatrics (Dermatology)
Feinberg School of Medicine
Northwestern University
Dr. Paller discusses the FDA approval of Dupixent® (dupilumab) for children aged 6 to 11 years with moderate-to-severe atopic dermatitis (eczema), whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable.
MedicalResearch.com: What is the background for this announcement? Would you briefly discuss what is meant by atopic dermatitis and how it affects children?
Response: “Atopic dermatitis, the most common form of eczema, is a chronic inflammatory disease that often appears as a rash on the skin. Moderate-to-severe atopic dermatitis is characterized by rashes that can potentially cover much of the body and can include intense, persistent itching, skin lesions and skin dryness, cracking, redness or darkness, crusting and oozing. Itch is one of the most burdensome symptoms for patients and can be debilitating.
This recent FDA approval expands the use of Dupilumab in the U.S. to include children aged 6 to 11 years with uncontrolled moderate-to-severe atopic dermatitis, making it the only biologic medicine approved for this use in this population. Dupilumab is also approved in the U.S. to treat patients aged 12 years and older with moderate-to-severe atopic dermatitis.
Moderate-to-severe atopic dermatitis can place a particularly substantial burden on young children aged 6 to 11 years and their families. Limited treatment options leave many of these children to cope with intense, unrelenting itch and skin lesions. Families of these children can spend countless hours helping them to manage their disease.”
Dr. Paller[/caption]
Amy S Paller, MD
Chair, Department of Dermatology
Director, Skin Biology and Diseases Resource-Based Center
Walter J. Hamlin Professor of Dermatology
Professor of Dermatology and Pediatrics (Dermatology)
Feinberg School of Medicine
Northwestern University
Dr. Paller discusses the FDA approval of Dupixent® (dupilumab) for children aged 6 to 11 years with moderate-to-severe atopic dermatitis (eczema), whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable.
MedicalResearch.com: What is the background for this announcement? Would you briefly discuss what is meant by atopic dermatitis and how it affects children?
Response: “Atopic dermatitis, the most common form of eczema, is a chronic inflammatory disease that often appears as a rash on the skin. Moderate-to-severe atopic dermatitis is characterized by rashes that can potentially cover much of the body and can include intense, persistent itching, skin lesions and skin dryness, cracking, redness or darkness, crusting and oozing. Itch is one of the most burdensome symptoms for patients and can be debilitating.
This recent FDA approval expands the use of Dupilumab in the U.S. to include children aged 6 to 11 years with uncontrolled moderate-to-severe atopic dermatitis, making it the only biologic medicine approved for this use in this population. Dupilumab is also approved in the U.S. to treat patients aged 12 years and older with moderate-to-severe atopic dermatitis.
Moderate-to-severe atopic dermatitis can place a particularly substantial burden on young children aged 6 to 11 years and their families. Limited treatment options leave many of these children to cope with intense, unrelenting itch and skin lesions. Families of these children can spend countless hours helping them to manage their disease.”





 Dr. Mishra[/caption]
Santosh K. Mishra M.Tech., PhD
Assistant Professor of Neuroscience
Department of Molecular Biomedical Sciences
NC State Veterinary Medicine
Raleigh, NC 2760
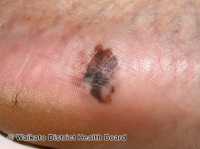
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by atopic dermatitis?
Response: Chronic allergic itch is a worldwide problem that leads to substantial health expenses,but what causes this universal urge to scratch remains elusive in chronic allergic itch. Atopic dermatitis is a common allergic skin disease that often associated with extremely itchy and inflamed skin.
In our study, we showed, for the first time, a molecular pathway that is involved in chronic allergic itch as we identified an endogenous mediator (periostin) and a new role for its sensory neuron receptor, the integrin αVβ3, which drives the excitability and transmission of itch signal to the spinal cord.
Dr. Mishra[/caption]
Santosh K. Mishra M.Tech., PhD
Assistant Professor of Neuroscience
Department of Molecular Biomedical Sciences
NC State Veterinary Medicine
Raleigh, NC 2760
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by atopic dermatitis?
Response: Chronic allergic itch is a worldwide problem that leads to substantial health expenses,but what causes this universal urge to scratch remains elusive in chronic allergic itch. Atopic dermatitis is a common allergic skin disease that often associated with extremely itchy and inflamed skin.
In our study, we showed, for the first time, a molecular pathway that is involved in chronic allergic itch as we identified an endogenous mediator (periostin) and a new role for its sensory neuron receptor, the integrin αVβ3, which drives the excitability and transmission of itch signal to the spinal cord. 
