Orthopedics / 05.05.2026
NEJM: Arthroscopic Partial Meniscectomy Provided No Benefit Over Placebo Surgery and May Worsen Longterm Outcomes
[caption id="attachment_73589" align="alignleft" width="200"] Dr. Järvinen[/caption]
MedicalResearch.com Interview with:
Teppo Järvinen MD PhD
Professor of Orthopaedics and Traumatology
University of Helsinki, HUS
MedicalResearch.com: What is the background for this study?
Response: Arthroscopic partial meniscectomy (APM) is one of the most frequently performed orthopedic procedures worldwide, particularly for middle-aged and older patients with knee pain and a degenerative meniscal tear.
Over the past 15–20 years, multiple randomized trials have questioned the effectiveness of this procedure, showing no meaningful benefit compared with non-operative care (typically, exercise or physical therapy) or even placebo (sham) surgery in the short to medium term.
Our study reports the 10-year follow-up of the FIDELITY trial, a randomized, placebo-surgery controlled trial.
Dr. Järvinen[/caption]
MedicalResearch.com Interview with:
Teppo Järvinen MD PhD
Professor of Orthopaedics and Traumatology
University of Helsinki, HUS
MedicalResearch.com: What is the background for this study?
Response: Arthroscopic partial meniscectomy (APM) is one of the most frequently performed orthopedic procedures worldwide, particularly for middle-aged and older patients with knee pain and a degenerative meniscal tear.
Over the past 15–20 years, multiple randomized trials have questioned the effectiveness of this procedure, showing no meaningful benefit compared with non-operative care (typically, exercise or physical therapy) or even placebo (sham) surgery in the short to medium term.
Our study reports the 10-year follow-up of the FIDELITY trial, a randomized, placebo-surgery controlled trial.
 Dr. Järvinen[/caption]
MedicalResearch.com Interview with:
Teppo Järvinen MD PhD
Professor of Orthopaedics and Traumatology
University of Helsinki, HUS
MedicalResearch.com: What is the background for this study?
Response: Arthroscopic partial meniscectomy (APM) is one of the most frequently performed orthopedic procedures worldwide, particularly for middle-aged and older patients with knee pain and a degenerative meniscal tear.
Over the past 15–20 years, multiple randomized trials have questioned the effectiveness of this procedure, showing no meaningful benefit compared with non-operative care (typically, exercise or physical therapy) or even placebo (sham) surgery in the short to medium term.
Our study reports the 10-year follow-up of the FIDELITY trial, a randomized, placebo-surgery controlled trial.
Dr. Järvinen[/caption]
MedicalResearch.com Interview with:
Teppo Järvinen MD PhD
Professor of Orthopaedics and Traumatology
University of Helsinki, HUS
MedicalResearch.com: What is the background for this study?
Response: Arthroscopic partial meniscectomy (APM) is one of the most frequently performed orthopedic procedures worldwide, particularly for middle-aged and older patients with knee pain and a degenerative meniscal tear.
Over the past 15–20 years, multiple randomized trials have questioned the effectiveness of this procedure, showing no meaningful benefit compared with non-operative care (typically, exercise or physical therapy) or even placebo (sham) surgery in the short to medium term.
Our study reports the 10-year follow-up of the FIDELITY trial, a randomized, placebo-surgery controlled trial.



 Prof. Lemesle[/caption]
Gilles Lemesle, M.D., Ph.D
Lille University Hospital, Lille, France
Prof. Lemesle[/caption]
Gilles Lemesle, M.D., Ph.D
Lille University Hospital, Lille, France




 Dr. Thomas Robert[/caption]
MedicalResearch.com Interview with:
Thomas Robert, MD, AIX
Associate Professor of Nephrology
APHM (Assistance Publique - Hopitaux de Marseille)
Marseille, Provence-Alpes-Côte d'Azur, France
[caption id="attachment_61498" align="alignleft" width="132"]
Dr. Thomas Robert[/caption]
MedicalResearch.com Interview with:
Thomas Robert, MD, AIX
Associate Professor of Nephrology
APHM (Assistance Publique - Hopitaux de Marseille)
Marseille, Provence-Alpes-Côte d'Azur, France
[caption id="attachment_61498" align="alignleft" width="132"] Dr. Letavernier[/caption]
Prof. Emmanuel Letavernier, MD PhD
Nephrologist at Tenon Hospital
Paris, France
MedicalResearch.com: What is the background for this study?
Response: Our work was prompted by emerging concerns surrounding the potential nephrotoxic effects of hair-straightening products containing glyoxylic acid. This inquiry was instigated by a patient who experienced three repeated acute episodes of kidney injury in June 2020, April 2021, and July 2022, each occurring shortly after a hair-straightening procedure. Notably, these episodes resolved with hydration.
Upon examining the composition of the hair product used by the patient, which contained glyoxylic acid, and considering the patient's report of painful ulcer scalp during application and subsequent scalp scarring, we suspected a potential link between exposure to glyoxylic acid and kidney injury. Consulting with my colleague, Professor Emmanuel Letavenier, a specialist in crystalline nephropathy at Paris, confirmed this suspicion.
In summer 2023, cases series have been reported by an Israeli team (
Dr. Letavernier[/caption]
Prof. Emmanuel Letavernier, MD PhD
Nephrologist at Tenon Hospital
Paris, France
MedicalResearch.com: What is the background for this study?
Response: Our work was prompted by emerging concerns surrounding the potential nephrotoxic effects of hair-straightening products containing glyoxylic acid. This inquiry was instigated by a patient who experienced three repeated acute episodes of kidney injury in June 2020, April 2021, and July 2022, each occurring shortly after a hair-straightening procedure. Notably, these episodes resolved with hydration.
Upon examining the composition of the hair product used by the patient, which contained glyoxylic acid, and considering the patient's report of painful ulcer scalp during application and subsequent scalp scarring, we suspected a potential link between exposure to glyoxylic acid and kidney injury. Consulting with my colleague, Professor Emmanuel Letavenier, a specialist in crystalline nephropathy at Paris, confirmed this suspicion.
In summer 2023, cases series have been reported by an Israeli team (

 Prof. Hiddo Lambers Heerspink, PhD PHARMD
Department of Clinical Pharmacy and Pharmacology
University Medical Center Groningen
Groningen
Prof. Hiddo Lambers Heerspink, PhD PHARMD
Department of Clinical Pharmacy and Pharmacology
University Medical Center Groningen
Groningen
 Dr. Greenhawt[/caption]
Matthew Greenhawt, MD, MBA, MSc
Professor of Pediatrics
Section of Allergy and Immunology
Director, Food Challenge and Research Unit
Children’s Hospital Colorado
University of Colorado School of Medicine
Anschutz Medical Campus
Dr. Greenhawt[/caption]
Matthew Greenhawt, MD, MBA, MSc
Professor of Pediatrics
Section of Allergy and Immunology
Director, Food Challenge and Research Unit
Children’s Hospital Colorado
University of Colorado School of Medicine
Anschutz Medical Campus



 Dr. Kim[/caption]
Brian S Kim, MD, MTR
Sol and Clara Kest Professor
Vice Chair of Research | Site Chair, Mount Sinai West and Morningside
Director, Mark Lebwohl Center for Neuroinflammation and Sensation
The Kimberly and Eric J. Waldman Department of Dermatology
Precision Immunology Institute | Friedman Brain Institute
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: We generated preliminary data in mice that difelikefalin suppresses itch robustly with very little to no effect on inflammation in a model of atopic dermatitis. This led us to hypothesize that the primary mode of action of difelikefalin must be by direct modulation of sensory neurons to suppress itch and thus would be best suited for a neuropathic itch condition. These are conditions in which itch occurs relatively independently of skin inflammation as in atopic dermatitis due to hyperactive nerves.
Dr. Kim[/caption]
Brian S Kim, MD, MTR
Sol and Clara Kest Professor
Vice Chair of Research | Site Chair, Mount Sinai West and Morningside
Director, Mark Lebwohl Center for Neuroinflammation and Sensation
The Kimberly and Eric J. Waldman Department of Dermatology
Precision Immunology Institute | Friedman Brain Institute
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: We generated preliminary data in mice that difelikefalin suppresses itch robustly with very little to no effect on inflammation in a model of atopic dermatitis. This led us to hypothesize that the primary mode of action of difelikefalin must be by direct modulation of sensory neurons to suppress itch and thus would be best suited for a neuropathic itch condition. These are conditions in which itch occurs relatively independently of skin inflammation as in atopic dermatitis due to hyperactive nerves.



 Dr. Han[/caption]
Meilan K Han MD, MS
Henry Sewall Professor of Medicine
Professor of Internal Medicine and Section Head
Division of Pulmonary and Critical Care Medicine, Medical School
University of Michigan
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In the NIH sponsored SPIROMICS study we demonstrated that symptomatic, tobacco exposed individuals have frequent exacerbations. Many of these individuals are treated with the same inhaled medications that have shown benefit in COPD, but we don’t have any evidence basis for this practice.
Dr. Han[/caption]
Meilan K Han MD, MS
Henry Sewall Professor of Medicine
Professor of Internal Medicine and Section Head
Division of Pulmonary and Critical Care Medicine, Medical School
University of Michigan
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In the NIH sponsored SPIROMICS study we demonstrated that symptomatic, tobacco exposed individuals have frequent exacerbations. Many of these individuals are treated with the same inhaled medications that have shown benefit in COPD, but we don’t have any evidence basis for this practice.



 Dr. Bailey[/caption]
Michael Bailey Ph.D.
Senior Principal Engineer, Applied Physics Laboratory
Associate Professor. Mechanical Engineering
Adjunct Associate Professor Urology
MedicalResearch.com: What is the background for this study?
Response: Small (< 6 mm) kidney stones are common and often are asymptomatic. Do you do surgery or wait for them to cause a problem? Or specifically here if you are getting surgery already for other stones that are causing a problem do you take the time and possibly extra risk of cleaning out the small stone in the kidney or in the other kidney?
Dr. Bailey[/caption]
Michael Bailey Ph.D.
Senior Principal Engineer, Applied Physics Laboratory
Associate Professor. Mechanical Engineering
Adjunct Associate Professor Urology
MedicalResearch.com: What is the background for this study?
Response: Small (< 6 mm) kidney stones are common and often are asymptomatic. Do you do surgery or wait for them to cause a problem? Or specifically here if you are getting surgery already for other stones that are causing a problem do you take the time and possibly extra risk of cleaning out the small stone in the kidney or in the other kidney?

 Dr. LeBoff[/caption]
Meryl S. LeBoff, MD
Dr. LeBoff[/caption]
Meryl S. LeBoff, MD
 Dr. Manson[/caption]
JoAnn E. Manson, MD, DrPH
Professor, Epidemiology, Harvard T.H. Chan School Of Public Health
Michael and Lee Bell Professor of Women's Health, Medicine, Harvard Medical School
Chief, Preventive Medicine, Brigham And Women's Hospital
Co-Director, Womens Health, Brigham And Women's Hospital
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Osteoporosis is a major public health problem. Although supplemental vitamin D has been widely used to reduce the risk of fractures in the general population, studies of the effects of vitamin D on fractures, the most important bone health outcome, have been conflicting.
Randomized controlled trials, the highest quality studies, from around the world have shown benefit, no effect, or even harm of supplemental vitamin D on risk of fractures. Some of the trials used bolus dosing, had small samples sizes or short study duration, and co-administered calcium. No large RCTS of this scale tested whether daily supplemental vitamin D (without co-administration with calcium) prevented fractures in the US population.
To fill these knowledge gaps, we tested the hypothesis in this ancillary study to VITAL, whether daily supplemental vitamin D3 reduced the risk of incident total, non-spine and hip fractures in women and men in the US.
Dr. Manson[/caption]
JoAnn E. Manson, MD, DrPH
Professor, Epidemiology, Harvard T.H. Chan School Of Public Health
Michael and Lee Bell Professor of Women's Health, Medicine, Harvard Medical School
Chief, Preventive Medicine, Brigham And Women's Hospital
Co-Director, Womens Health, Brigham And Women's Hospital
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Osteoporosis is a major public health problem. Although supplemental vitamin D has been widely used to reduce the risk of fractures in the general population, studies of the effects of vitamin D on fractures, the most important bone health outcome, have been conflicting.
Randomized controlled trials, the highest quality studies, from around the world have shown benefit, no effect, or even harm of supplemental vitamin D on risk of fractures. Some of the trials used bolus dosing, had small samples sizes or short study duration, and co-administered calcium. No large RCTS of this scale tested whether daily supplemental vitamin D (without co-administration with calcium) prevented fractures in the US population.
To fill these knowledge gaps, we tested the hypothesis in this ancillary study to VITAL, whether daily supplemental vitamin D3 reduced the risk of incident total, non-spine and hip fractures in women and men in the US.


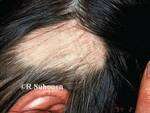
 Brett King, MD, PhD, FAAD
Associate Professor of Dermatology
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Alopecia areata is an autoimmune disorder marked by disfiguring, non-scarring hair loss, and there are no therapies approved by the U.S. Food and Drug Administration for treatment of the disease. JAK inhibitors are showing promise for treatment of severe alopecia areata. In this work, the pooled results of two phase 3 clinical trials of the JAK inhibitor baricitinib were reported out to 52 weeks.
Brett King, MD, PhD, FAAD
Associate Professor of Dermatology
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Alopecia areata is an autoimmune disorder marked by disfiguring, non-scarring hair loss, and there are no therapies approved by the U.S. Food and Drug Administration for treatment of the disease. JAK inhibitors are showing promise for treatment of severe alopecia areata. In this work, the pooled results of two phase 3 clinical trials of the JAK inhibitor baricitinib were reported out to 52 weeks.
