ASCO, AstraZeneca, Author Interviews, Cancer Research, Ovarian Cancer / 02.06.2020
LYNPARZA Demonstrates Improved Overall Survival in BRCA-Mutated, Platinum-Sensitive Relapsed Ovarian Cancer
MedicalResearch.com Interview with:
[caption id="attachment_54434" align="alignleft" width="200"] Mark Sims[/caption]
Mark Sims
US Franchise Head
Women’s Cancer & DNA Damage Response
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: SOLO2 is a Phase III, randomized, double-blind, multicenter trial designed to determine the efficacy and safety of LYNPARZA tablets as a maintenance monotherapy compared with placebo, in patients with platinum-sensitive relapsed or recurrent gBRCA-mutated (BRCAm) ovarian cancer. The trial included 295 patients with germline BRCA1 or BRCA2 mutations who had received at least 2 prior lines of platinum-based chemotherapy and were in complete or partial response. The trial met its primary endpoint in October 2016, showing maintenance treatment with LYNPARZA significantly improved progression-free survival to a median of 19.1 months vs 5.5 months with placebo (a hazard ratio of 0.30; a 95% confidence interval of 0.22 to 0.41; a p value of <0.0001).
Mark Sims[/caption]
Mark Sims
US Franchise Head
Women’s Cancer & DNA Damage Response
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: SOLO2 is a Phase III, randomized, double-blind, multicenter trial designed to determine the efficacy and safety of LYNPARZA tablets as a maintenance monotherapy compared with placebo, in patients with platinum-sensitive relapsed or recurrent gBRCA-mutated (BRCAm) ovarian cancer. The trial included 295 patients with germline BRCA1 or BRCA2 mutations who had received at least 2 prior lines of platinum-based chemotherapy and were in complete or partial response. The trial met its primary endpoint in October 2016, showing maintenance treatment with LYNPARZA significantly improved progression-free survival to a median of 19.1 months vs 5.5 months with placebo (a hazard ratio of 0.30; a 95% confidence interval of 0.22 to 0.41; a p value of <0.0001).
 Mark Sims[/caption]
Mark Sims
US Franchise Head
Women’s Cancer & DNA Damage Response
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: SOLO2 is a Phase III, randomized, double-blind, multicenter trial designed to determine the efficacy and safety of LYNPARZA tablets as a maintenance monotherapy compared with placebo, in patients with platinum-sensitive relapsed or recurrent gBRCA-mutated (BRCAm) ovarian cancer. The trial included 295 patients with germline BRCA1 or BRCA2 mutations who had received at least 2 prior lines of platinum-based chemotherapy and were in complete or partial response. The trial met its primary endpoint in October 2016, showing maintenance treatment with LYNPARZA significantly improved progression-free survival to a median of 19.1 months vs 5.5 months with placebo (a hazard ratio of 0.30; a 95% confidence interval of 0.22 to 0.41; a p value of <0.0001).
Mark Sims[/caption]
Mark Sims
US Franchise Head
Women’s Cancer & DNA Damage Response
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: SOLO2 is a Phase III, randomized, double-blind, multicenter trial designed to determine the efficacy and safety of LYNPARZA tablets as a maintenance monotherapy compared with placebo, in patients with platinum-sensitive relapsed or recurrent gBRCA-mutated (BRCAm) ovarian cancer. The trial included 295 patients with germline BRCA1 or BRCA2 mutations who had received at least 2 prior lines of platinum-based chemotherapy and were in complete or partial response. The trial met its primary endpoint in October 2016, showing maintenance treatment with LYNPARZA significantly improved progression-free survival to a median of 19.1 months vs 5.5 months with placebo (a hazard ratio of 0.30; a 95% confidence interval of 0.22 to 0.41; a p value of <0.0001).



 Dr. Schünemann[/caption]
Holger Schünemann, MD, PhD, FRCPC
Professor of Clinical Epidemiology and of Medicine
Co-Director, WHO Collaborating Centre for Infectious Diseases,
Research Methods and Recommendations
Director, Cochrane Canada and McMaster GRADE Centre
Department of Health Research Methods, Evidence, and Impact
Canada
MedicalResearch.com: What is the background for this study?
Response: Many countries and regions have issued conflicting advice about physical distancing to reduce transmission of COVID-19, based on limited information. In addition, the questions of whether masks and eye coverings might reduce transmission of COVID-19 in the general population, and what the optimum use of masks in healthcare settings is, have been debated during the pandemic.
Dr. Schünemann[/caption]
Holger Schünemann, MD, PhD, FRCPC
Professor of Clinical Epidemiology and of Medicine
Co-Director, WHO Collaborating Centre for Infectious Diseases,
Research Methods and Recommendations
Director, Cochrane Canada and McMaster GRADE Centre
Department of Health Research Methods, Evidence, and Impact
Canada
MedicalResearch.com: What is the background for this study?
Response: Many countries and regions have issued conflicting advice about physical distancing to reduce transmission of COVID-19, based on limited information. In addition, the questions of whether masks and eye coverings might reduce transmission of COVID-19 in the general population, and what the optimum use of masks in healthcare settings is, have been debated during the pandemic.


 Dr. Kao-Ping Chua[/caption]
Kao-Ping Chua, M.D., Ph.D.
Assistant Professor, Pediatrics, Medical School
Susan B. Meister Child Health Evaluation and Research Center
University of Michigan
MedicalResearch.com: What is the background for this study?
Response: Due to high and rising prices, insulin has become increasingly unaffordable for patients with type 1 diabetes who must pay out-of-pocket for this life-saving medication. Over the past 5 months, many states and insurers have taken steps to cap insulin out-of-pocket spending. For example, Cigna imposed a $25 monthly cap earlier this year. This week, the Centers for Medicare and Medicare Services announced a $35 monthly cap for many Medicare Part D beneficiaries.
Dr. Kao-Ping Chua[/caption]
Kao-Ping Chua, M.D., Ph.D.
Assistant Professor, Pediatrics, Medical School
Susan B. Meister Child Health Evaluation and Research Center
University of Michigan
MedicalResearch.com: What is the background for this study?
Response: Due to high and rising prices, insulin has become increasingly unaffordable for patients with type 1 diabetes who must pay out-of-pocket for this life-saving medication. Over the past 5 months, many states and insurers have taken steps to cap insulin out-of-pocket spending. For example, Cigna imposed a $25 monthly cap earlier this year. This week, the Centers for Medicare and Medicare Services announced a $35 monthly cap for many Medicare Part D beneficiaries.


 Dr. Meiri[/caption]
Amir Meiri, MD MPH
Atrius Health/Department of Population Medicine (DPM) | Delivery System Science Fellow
HMS and HPHCI, DPM | General Internal Medicine Fellow
Atrius Health Kenmore | Urgent Care Physician
VA Boston Healthcare | Attending in Internal Medicine and Emergency Medicine
MedicalResearch.com: What is the background for this study?
Response: There has been significant media reporting about rising insulin prices and the health impacts of those exorbitant prices. However, it was not clear how these insulin prices may impact out-of-pocket costs among commercially insured patients; though it is clear that those without insurance are affected per previous media reports. Our study examines the difference between insulin manufacturer-set prices and what patients actually pay, the out-of-pocket cost, in the context of the type of insurance patients have.
Dr. Meiri[/caption]
Amir Meiri, MD MPH
Atrius Health/Department of Population Medicine (DPM) | Delivery System Science Fellow
HMS and HPHCI, DPM | General Internal Medicine Fellow
Atrius Health Kenmore | Urgent Care Physician
VA Boston Healthcare | Attending in Internal Medicine and Emergency Medicine
MedicalResearch.com: What is the background for this study?
Response: There has been significant media reporting about rising insulin prices and the health impacts of those exorbitant prices. However, it was not clear how these insulin prices may impact out-of-pocket costs among commercially insured patients; though it is clear that those without insurance are affected per previous media reports. Our study examines the difference between insulin manufacturer-set prices and what patients actually pay, the out-of-pocket cost, in the context of the type of insurance patients have.



 CDC PHIL image[/caption]
MedicalResearch.com: What is the background for this study?
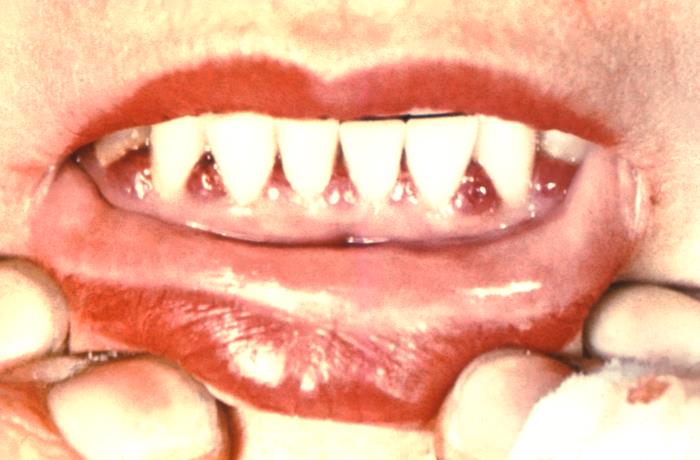
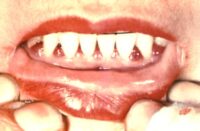
Response: This report reviewed three measures of tooth loss among adults over 50 years old with selected chronic conditions. The report analyzed data from the 2011–2016 cycles of the National Health and Nutrition Examination Survey (NHANES) to compare the rates of total tooth loss, severe tooth loss (less than 8 teeth), and lacking functional dentition (less than 20 teeth) among people with and without selected chronic conditions.
CDC PHIL image[/caption]
MedicalResearch.com: What is the background for this study?
Response: This report reviewed three measures of tooth loss among adults over 50 years old with selected chronic conditions. The report analyzed data from the 2011–2016 cycles of the National Health and Nutrition Examination Survey (NHANES) to compare the rates of total tooth loss, severe tooth loss (less than 8 teeth), and lacking functional dentition (less than 20 teeth) among people with and without selected chronic conditions.














 Dr. Filbey[/caption]
Francesca Filbey, PhD
Associate Provost and Professor of Cognition and Neuroscience
Bert Moore Chair
The University of Texas at Dallas
MedicalResearch.com: What is the background for this study?
Response: Studies have reported differences in how males and females respond to cannabis and how they develop problems related to cannabis use. We sought to determine whether craving may underlie this difference in male and female cannabis users.
Dr. Filbey[/caption]
Francesca Filbey, PhD
Associate Provost and Professor of Cognition and Neuroscience
Bert Moore Chair
The University of Texas at Dallas
MedicalResearch.com: What is the background for this study?
Response: Studies have reported differences in how males and females respond to cannabis and how they develop problems related to cannabis use. We sought to determine whether craving may underlie this difference in male and female cannabis users.

