MedicalResearch.com Interview with:
[caption id="attachment_53839" align="alignleft" width="137"] Dr. Bahji[/caption]
Anees Bahji, MD
PGY5, Department of Psychiatry, Queen’s University
M.Sc. Candidate, Department of Public Health Sciences,
Queen’s University
Kingston, ON, Canada
MedicalResearch.com: What is the background for this study?
Dr. Bahji[/caption]
Anees Bahji, MD
PGY5, Department of Psychiatry, Queen’s University
M.Sc. Candidate, Department of Public Health Sciences,
Queen’s University
Kingston, ON, Canada
MedicalResearch.com: What is the background for this study?
 Dr. Bahji[/caption]
Anees Bahji, MD
PGY5, Department of Psychiatry, Queen’s University
M.Sc. Candidate, Department of Public Health Sciences,
Queen’s University
Kingston, ON, Canada
MedicalResearch.com: What is the background for this study?
Dr. Bahji[/caption]
Anees Bahji, MD
PGY5, Department of Psychiatry, Queen’s University
M.Sc. Candidate, Department of Public Health Sciences,
Queen’s University
Kingston, ON, Canada
MedicalResearch.com: What is the background for this study?
- There has been much speculation into the existence of a withdrawal syndrome involving cannabis much like withdrawal syndromes from opioids or alcohol. Our goal for this study was to estimate the prevalence of cannabis withdrawal syndrome (CWS) and to identify any risk factors for CWS.
- There has been a lot of research into cannabis withdrawal syndrome (CWS) in the past. A big part of this review involved understanding where the CWS field is in terms of the shared understanding on its epidemiology and physiology.
- To that end, we were not surprised to find that the prevalence of cannabis withdrawal syndrome was high. However, we found that some of the characteristics of CWS are consistent with other substance use disorders, which really serves to legitimize the decision to classify CWS and cannabis use disorders as psychiatric conditions.







![MedicalResearch.com Interview with: Melissa Bondy, PhD Chair, Department of Epidemiology and Population Health Stanford University MedicalResearch.com: What is the background for this study? Response: Faculty researchers in Stanford’s Department of Epidemiology & Population Health and collaborators from Baylor College of Medicine and Stanford’s Department of Dermatology developed an online survey aimed at rapidly assessing public concerns about the COVID-19 crisis. This survey, which was posted on 3 social media platforms (Twitter, Facebook, and Nextdoor) on March 14, 2020, collected invaluable data about COVID-19, including symptoms, concerns, and individual actions taken by respondents. Twitter and Facebook posts were sharable to facilitate snowball sampling. The survey was comprised of 21 (multiple-choice, single-choice, numeric, and open-ended) questions, which were designed to collect data concerning respondent demographics and recent cold and flu-like illnesses (if any), as well as information about participants’ concerns and any lifestyle changes that occurred as a result of the COVID-19 crisis. All questions were optional, so response rates were variable. MedicalResearch.com: What are the main findings? Response: 9009 surveys were completed within a 48 hour period, between March 14 and 16. A majority were very or extremely concerns about COVID-19, though concern varied by age. Most respondents reported making lifestyle changes in response to COVID-19. The most common changes were more hand washing, avoiding social gatherings, and stockpiling food and supplies. About half of respondents were self-isolating most of the time before shelter-in-place orders, and leaving the house only to buy food and essentials. Respondents who were not self-isolating all of the time, before the shelter-in-place order, most commonly listed not being able to afford missing work as the reason. The top concerns reported by survey participants were getting sick because of COVID-19 and not being able to get medical care. The most common difficulties reported were obtaining hand sanitizer, food, and childcare. MedicalResearch.com: What should readers take away from your report? Response: This convenience sample survey distributed in mid-March 2020 across 3 social media platforms reflect respondents’ serious concerns about COVID-19. They were preparing for the crisis by washing hands, isolating at home, and practicing social distancing to the best of their abilities. Levels of concern about COVID-19 varied by age groups. Participants expressed the most common difficulties included getting sanitizer or food, arranging childcare, and lost wages or reduced working hours. MedicalResearch.com: What recommendations do you have for future research as a result of this work? Response: Groups who are generally known to be underrepresented in social media, particularly men and older individuals, were underrepresented among survey participants. Few respondents reported concerns about their employment, but the study did not collect information about participants’ socioeconomic status (and was likely high). Our future work will expand geographic representation among survey respondents, with greater focus on longitudinal trends on the health, financial, and social concerns in the United States regarding the COVID-19 pandemic. No disclosures or conflicts of interest. Citation: Nelson LM, Simard JF, Oluyomi A, et al. US Public Concerns About the COVID-19 Pandemic From Results of a Survey Given via Social Media. JAMA Intern Med. Published online April 07, 2020. doi:10.1001/jamainternmed.2020.1369 https://jamanetwork.com/journals/jamainternalmedicine/fullarticle/2764368?guestAccessKey=cd7f419d-b177-44d2-86b2-c3e0254b3152&utm_source=For_The_Media&utm_medium=referral&utm_campaign=ftm_links&utm_content=tfl&utm_term=040720 [subscribe] Last Modified: [last-modified] The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2020/04/Melissa-Bondy.jpg)



 Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
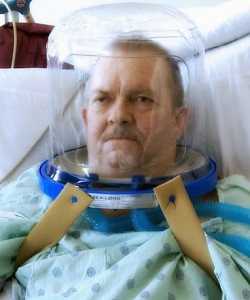
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.
Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.




















 Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.
Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.