Author Interviews, Diabetes, Supplements / 08.01.2020
Could a Ketone Drink Help Keep Blood Sugar Under Control?
MedicalResearch.com Interview with:
[caption id="attachment_52710" align="alignleft" width="200"] Dr. Little[/caption]
Jonathan P. Little PhD
Associate Professor Canadian Institutes of Health Research New Investigator
Michael Smith Foundation for Health Research Scholar
Faculty of Health and Social Development
School of Health and Exercise Sciences
The University of British Columbia Syilx Okanagan Nation Territory
Kelowna, BC Canada
MedicalResearch.com: What is the background for this study?
Response: Ketones are energy-yielding molecules that are bodies naturally produce during periods of starvation or when we restrict carbohydrate intake. Recently, scientists from Oxford and the NIH have created exogenous ketone supplements, which now enable us to be able to drink ketones. This puts our body into a unique state – we can consume a drink that raises blood levels of ketones without having to starve or restrict carbohydrate intake. Some are even touting ketone supplements as a “fourth macronutrient”.
Ketone supplements are primarily marketed for athletes to provide an alternative fuel for improving endurance exercise performance. We were actually studying how ketone supplements impacted exercise performance when we noticed that they consistently lowered blood glucose after participants consumed them. We went to the literature and found some classic papers where it was shown that infusing ketones did in fact lower glucose and the mechanism seemed to involve reducing liver glucose output. This was very exciting to us because we also study type 2 diabetes, a condition where blood sugars are too high and elevated liver glucose output is one of the major reasons. So we came up with the hypothesis that ketone supplements might be a unique strategy to help with blood glucose control.
In the recent study, we tested this out in a randomized crossover experiment in 15 participants with overweight/obesity who were at risk for type 2 diabetes. Participants drank the ketone monoester supplement or a placebo and 30 minutes later they consumed an oral glucose tolerance test drink containing 75 grams of sugar. Blood samples were collected for 2 hours after the glucose test drink.
Dr. Little[/caption]
Jonathan P. Little PhD
Associate Professor Canadian Institutes of Health Research New Investigator
Michael Smith Foundation for Health Research Scholar
Faculty of Health and Social Development
School of Health and Exercise Sciences
The University of British Columbia Syilx Okanagan Nation Territory
Kelowna, BC Canada
MedicalResearch.com: What is the background for this study?
Response: Ketones are energy-yielding molecules that are bodies naturally produce during periods of starvation or when we restrict carbohydrate intake. Recently, scientists from Oxford and the NIH have created exogenous ketone supplements, which now enable us to be able to drink ketones. This puts our body into a unique state – we can consume a drink that raises blood levels of ketones without having to starve or restrict carbohydrate intake. Some are even touting ketone supplements as a “fourth macronutrient”.
Ketone supplements are primarily marketed for athletes to provide an alternative fuel for improving endurance exercise performance. We were actually studying how ketone supplements impacted exercise performance when we noticed that they consistently lowered blood glucose after participants consumed them. We went to the literature and found some classic papers where it was shown that infusing ketones did in fact lower glucose and the mechanism seemed to involve reducing liver glucose output. This was very exciting to us because we also study type 2 diabetes, a condition where blood sugars are too high and elevated liver glucose output is one of the major reasons. So we came up with the hypothesis that ketone supplements might be a unique strategy to help with blood glucose control.
In the recent study, we tested this out in a randomized crossover experiment in 15 participants with overweight/obesity who were at risk for type 2 diabetes. Participants drank the ketone monoester supplement or a placebo and 30 minutes later they consumed an oral glucose tolerance test drink containing 75 grams of sugar. Blood samples were collected for 2 hours after the glucose test drink.
 Dr. Little[/caption]
Jonathan P. Little PhD
Associate Professor Canadian Institutes of Health Research New Investigator
Michael Smith Foundation for Health Research Scholar
Faculty of Health and Social Development
School of Health and Exercise Sciences
The University of British Columbia Syilx Okanagan Nation Territory
Kelowna, BC Canada
MedicalResearch.com: What is the background for this study?
Response: Ketones are energy-yielding molecules that are bodies naturally produce during periods of starvation or when we restrict carbohydrate intake. Recently, scientists from Oxford and the NIH have created exogenous ketone supplements, which now enable us to be able to drink ketones. This puts our body into a unique state – we can consume a drink that raises blood levels of ketones without having to starve or restrict carbohydrate intake. Some are even touting ketone supplements as a “fourth macronutrient”.
Ketone supplements are primarily marketed for athletes to provide an alternative fuel for improving endurance exercise performance. We were actually studying how ketone supplements impacted exercise performance when we noticed that they consistently lowered blood glucose after participants consumed them. We went to the literature and found some classic papers where it was shown that infusing ketones did in fact lower glucose and the mechanism seemed to involve reducing liver glucose output. This was very exciting to us because we also study type 2 diabetes, a condition where blood sugars are too high and elevated liver glucose output is one of the major reasons. So we came up with the hypothesis that ketone supplements might be a unique strategy to help with blood glucose control.
In the recent study, we tested this out in a randomized crossover experiment in 15 participants with overweight/obesity who were at risk for type 2 diabetes. Participants drank the ketone monoester supplement or a placebo and 30 minutes later they consumed an oral glucose tolerance test drink containing 75 grams of sugar. Blood samples were collected for 2 hours after the glucose test drink.
Dr. Little[/caption]
Jonathan P. Little PhD
Associate Professor Canadian Institutes of Health Research New Investigator
Michael Smith Foundation for Health Research Scholar
Faculty of Health and Social Development
School of Health and Exercise Sciences
The University of British Columbia Syilx Okanagan Nation Territory
Kelowna, BC Canada
MedicalResearch.com: What is the background for this study?
Response: Ketones are energy-yielding molecules that are bodies naturally produce during periods of starvation or when we restrict carbohydrate intake. Recently, scientists from Oxford and the NIH have created exogenous ketone supplements, which now enable us to be able to drink ketones. This puts our body into a unique state – we can consume a drink that raises blood levels of ketones without having to starve or restrict carbohydrate intake. Some are even touting ketone supplements as a “fourth macronutrient”.
Ketone supplements are primarily marketed for athletes to provide an alternative fuel for improving endurance exercise performance. We were actually studying how ketone supplements impacted exercise performance when we noticed that they consistently lowered blood glucose after participants consumed them. We went to the literature and found some classic papers where it was shown that infusing ketones did in fact lower glucose and the mechanism seemed to involve reducing liver glucose output. This was very exciting to us because we also study type 2 diabetes, a condition where blood sugars are too high and elevated liver glucose output is one of the major reasons. So we came up with the hypothesis that ketone supplements might be a unique strategy to help with blood glucose control.
In the recent study, we tested this out in a randomized crossover experiment in 15 participants with overweight/obesity who were at risk for type 2 diabetes. Participants drank the ketone monoester supplement or a placebo and 30 minutes later they consumed an oral glucose tolerance test drink containing 75 grams of sugar. Blood samples were collected for 2 hours after the glucose test drink.








 Dr. Anderson[/caption]
MedicalResearch.com Interview with:
Matilda Anderson MBBS MBS
General Surgery Trainee/Public Health/Researcher
Footscray, Victoria, Australia
MedicalResearch.com: What is the background for this study?
Response: Female representation in the surgical workforce is increasing. Previous studies have shown higher infertility rates and adverse pregnancy outcomes in this population. We aimed to accurately define the available research in this area and provide some basic recommendations about how workplaces can support their female surgical residents and surgeons.
On a more personal note- Dr Anderson is a female surgical resident and have seen countless pregnant colleagues remove themselves from operating rooms with the concern about how the conditions may affect their pregnancies. Dr. Anderson met Associate Professor Goldman at Harvard University (a leading expert on occupational reproductive hazards) and collaborated to explore this area further.
Dr. Anderson[/caption]
MedicalResearch.com Interview with:
Matilda Anderson MBBS MBS
General Surgery Trainee/Public Health/Researcher
Footscray, Victoria, Australia
MedicalResearch.com: What is the background for this study?
Response: Female representation in the surgical workforce is increasing. Previous studies have shown higher infertility rates and adverse pregnancy outcomes in this population. We aimed to accurately define the available research in this area and provide some basic recommendations about how workplaces can support their female surgical residents and surgeons.
On a more personal note- Dr Anderson is a female surgical resident and have seen countless pregnant colleagues remove themselves from operating rooms with the concern about how the conditions may affect their pregnancies. Dr. Anderson met Associate Professor Goldman at Harvard University (a leading expert on occupational reproductive hazards) and collaborated to explore this area further.


 Dr. Kistler[/caption]
Professor Peter M Kistler MBBS, PhD, FRACP
Head of Clinical Electrophysiology Research
Baker Heart and Diabetes Institute
Head of Electrophysiology at The Alfred hospital
Professor of Medicine
University of Melbourne.
MedicalResearch.com: What is the background for this study?
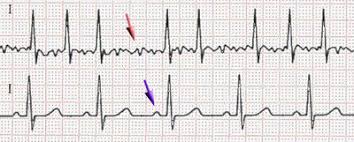
Response: There is a well known association between alcohol intake and atrial fibrillation form population based studies which demonstrate that for every 1 standard drink the incidence of AFib increases by 8%.
This is the first randomised study to determine of alcohol reduction/abstinence leads to a reduction in AFib episodes and time to recurrence.
Dr. Kistler[/caption]
Professor Peter M Kistler MBBS, PhD, FRACP
Head of Clinical Electrophysiology Research
Baker Heart and Diabetes Institute
Head of Electrophysiology at The Alfred hospital
Professor of Medicine
University of Melbourne.
MedicalResearch.com: What is the background for this study?
Response: There is a well known association between alcohol intake and atrial fibrillation form population based studies which demonstrate that for every 1 standard drink the incidence of AFib increases by 8%.
This is the first randomised study to determine of alcohol reduction/abstinence leads to a reduction in AFib episodes and time to recurrence.