Yes to Performance and No to Doping: Is Hemp Extract Legal in Sports?
Hemp-derived cannabidiol (CBD), or hemp extract, has been gaining in popularity in recent years. As long as CBD extract is produced using hemp plants, not marijuana, and contains less than 0.3% THC, it's considered legal in all 50 states. Some states enforce restrictions on buyers, but federally, general consumers who want to use CBD are in the clear.
Unfortunately, while the social stigma surrounding hemp extract has largely been eradicated in general populations, the athletic community hasn't fully embraced the benefits of CBD and some competitive fields still have strict regulations in place governing its use. Read on to find out what amateur, college, and professional athletes need to know.
CBD for Amateur Athletes
Amateur athletes don't have to worry about the restrictions put in place by regulatory bodies. They are free to take advantage of CBD's non-psychoactive therapeutic benefits, which include reduced joint pain and inflammation and improved muscle recovery after strenuous workouts. Guardian Athletic has more information about the benefits of cannabidiol for athletes looking to reduce recovery time and improve performance on their website.
Those who plan to engage in collegiate athletics or try out for semi-pro or professional sports teams should be aware that CBD can show up on a blood test for several days after consumption. They should also note that some hemp extracts contain up to 0.3% THC. This extremely low concentration is unlikely to produce a positive test result for marijuana, but it's still better to buy CBD from a reputable supplier that uses lab testing to ensure 0% THC concentrations.
FASENRA® – Benralizumab for Adolescent Patients with Severe, Uncontrolled Asthma
 Dr. Martin[/caption]
Ubaldo Martin MD
VP Clinical Respiratory
RIA Late Stage Development
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: BORA was an extension study evaluating the long-term safety and specific aspects of efficacy in patients who had previously been in the benralizumab pivotal studies. After the patients completed the pivotal studies (Calima, Sirocco and Zonda), they were eligible to join BORA which followed adults for an additional year and adolescent for an additional 2 years. All patients receive one of two dosages of benralizumab. The abstract reports the outcomes of adolescents in the BORA study who were followed for approximately 3 years in total.
Dr. Martin[/caption]
Ubaldo Martin MD
VP Clinical Respiratory
RIA Late Stage Development
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: BORA was an extension study evaluating the long-term safety and specific aspects of efficacy in patients who had previously been in the benralizumab pivotal studies. After the patients completed the pivotal studies (Calima, Sirocco and Zonda), they were eligible to join BORA which followed adults for an additional year and adolescent for an additional 2 years. All patients receive one of two dosages of benralizumab. The abstract reports the outcomes of adolescents in the BORA study who were followed for approximately 3 years in total.
Trulance® (plecanatide) For Chronic Idiopathic Constipation and Irritable Bowel Syndrome-C
 Dr. Sayuk[/caption]
Gregory S. Sayuk, MD, MPH
John T. Milliken Department of Medicine
Division of Gastroenterology
Associate Professor of Medicine and Psychiatry
Associate Director, Fellowship Training Program
Washington University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: The primary objective of this analysis, “Plecanatide for Patients with Chronic Idiopathic Constipation and Irritable Bowel Syndrome‒Constipation: Analysis of Abdominal Pain from Four Randomized Phase 3 Clinical Trials,” was to determine the impact of plecanatide on abdominal pain in patients with chronic idiopathic constipation (CIC) and irritable bowel syndrome with constipation (IBS-C), in addition to comparing efficacy results in patient populations with minimal to mild pain versus moderate to severe pain at baseline. This was a post hoc analysis of data from the four large, phase 3 plecanatide pivotal trials in CIC and IBS-C.
Dr. Sayuk[/caption]
Gregory S. Sayuk, MD, MPH
John T. Milliken Department of Medicine
Division of Gastroenterology
Associate Professor of Medicine and Psychiatry
Associate Director, Fellowship Training Program
Washington University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: The primary objective of this analysis, “Plecanatide for Patients with Chronic Idiopathic Constipation and Irritable Bowel Syndrome‒Constipation: Analysis of Abdominal Pain from Four Randomized Phase 3 Clinical Trials,” was to determine the impact of plecanatide on abdominal pain in patients with chronic idiopathic constipation (CIC) and irritable bowel syndrome with constipation (IBS-C), in addition to comparing efficacy results in patient populations with minimal to mild pain versus moderate to severe pain at baseline. This was a post hoc analysis of data from the four large, phase 3 plecanatide pivotal trials in CIC and IBS-C.RNA Profiling Identifies Distinct Classes of White Fat Cells
 Dr. Kahn[/caption]
Ronald Kahn, MD
Chief Academic Officer, Joslin Diabetes Center
Mary K. Iacocca Professor of Medicine
Harvard Medical School
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Adipose tissue is a heterogeneous organ and composed of several cell types, including mature adipocytes, preadipocytes, stem cells, endothelial cells, and various blood cells.
Different adipose depots have distinct physiological functions associated with their anatomical location and cell composition. For example, accumulation of intra-abdominal (visceral) white adipose tissue is associated with insulin resistance and metabolic syndrome, whereas accumulation of subcutaneous adipose tissue is not metabolically detrimental and may be even associated with increased insulin sensitivity.
Determining the mechanisms for these phenotypic differences could lead to development of novel therapies for diabetes, obesity, and their associated morbidities.
A central challenging question in research of metabolic disease is whether disease risk for diabetes and metabolic syndrome is driven by a subset of fat cells that may interact with environmental stresses in disease pathogenesis in a way different from other fat cells. Indeed, previous studies from the Kahn lab have shown different fat cells in a single depot from the mouse may exhibit developmental heterogeneity.
In this new study, we attempted to address this question for human white fat using a synergistic application of several methodologies:
1) single cell transcriptional profiling of human white fat during differentiation,
2) analysis of individual clones of white fat cells taken from humans at surgery,
3) novel computer based network analysis and
4) integration of the gene signatures across experimental models. Single-cell RNA sequencing is an ideal technique to profile gene expression of heterogeneous cell populations obtained from a single tissue, including fat tissue.
Dr. Kahn[/caption]
Ronald Kahn, MD
Chief Academic Officer, Joslin Diabetes Center
Mary K. Iacocca Professor of Medicine
Harvard Medical School
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Adipose tissue is a heterogeneous organ and composed of several cell types, including mature adipocytes, preadipocytes, stem cells, endothelial cells, and various blood cells.
Different adipose depots have distinct physiological functions associated with their anatomical location and cell composition. For example, accumulation of intra-abdominal (visceral) white adipose tissue is associated with insulin resistance and metabolic syndrome, whereas accumulation of subcutaneous adipose tissue is not metabolically detrimental and may be even associated with increased insulin sensitivity.
Determining the mechanisms for these phenotypic differences could lead to development of novel therapies for diabetes, obesity, and their associated morbidities.
A central challenging question in research of metabolic disease is whether disease risk for diabetes and metabolic syndrome is driven by a subset of fat cells that may interact with environmental stresses in disease pathogenesis in a way different from other fat cells. Indeed, previous studies from the Kahn lab have shown different fat cells in a single depot from the mouse may exhibit developmental heterogeneity.
In this new study, we attempted to address this question for human white fat using a synergistic application of several methodologies:
1) single cell transcriptional profiling of human white fat during differentiation,
2) analysis of individual clones of white fat cells taken from humans at surgery,
3) novel computer based network analysis and
4) integration of the gene signatures across experimental models. Single-cell RNA sequencing is an ideal technique to profile gene expression of heterogeneous cell populations obtained from a single tissue, including fat tissue.
USPSTF: Recommendations for Prevention and Cessation of Tobacco Use in Children and Adolescents
 Dr. Silverstein[/caption]
Dr. Michael Silverstein M.D., M.P.H
Professor of Pediatrics
Director of the Division of General Academic Pediatrics
Vice Chair of Research, Department of Pediatrics
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: The number of children and teens who use tobacco products continues to be a major issue in the U.S., driven largely by an increase in e-cigarette use, which makes preventing tobacco use among young people critical to the health of our nation. To help prevent kids and teens from starting to use tobacco, the Task Force recommends clinicians provide behavioral interventions, such as education or brief counseling.
Dr. Silverstein[/caption]
Dr. Michael Silverstein M.D., M.P.H
Professor of Pediatrics
Director of the Division of General Academic Pediatrics
Vice Chair of Research, Department of Pediatrics
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: The number of children and teens who use tobacco products continues to be a major issue in the U.S., driven largely by an increase in e-cigarette use, which makes preventing tobacco use among young people critical to the health of our nation. To help prevent kids and teens from starting to use tobacco, the Task Force recommends clinicians provide behavioral interventions, such as education or brief counseling. Cancer Drug Approval: FDA Acceptance of Surrogate End Points
 Dr. Chen[/caption]
Emerson Y. Chen, MD
Assistant Professor of Medicine, Medical Oncology
Knight Cancer Institute, Oregon Health & Science University
Portland, OR 97239
MedicalResearch.com: What is the background for this study?
Response: Our research group had previously studied how oncology drugs are approved in these two previous papers listed below. One is focused on the time delay trade-off from surrogate endpoints (i.e. response rate and progression-free survival) over definite endpoints (i.e. overall survival and quality of life). The other is focused on how promising the response rate of a drug candidate have to be to be considered for oncology drug approval.
Dr. Chen[/caption]
Emerson Y. Chen, MD
Assistant Professor of Medicine, Medical Oncology
Knight Cancer Institute, Oregon Health & Science University
Portland, OR 97239
MedicalResearch.com: What is the background for this study?
Response: Our research group had previously studied how oncology drugs are approved in these two previous papers listed below. One is focused on the time delay trade-off from surrogate endpoints (i.e. response rate and progression-free survival) over definite endpoints (i.e. overall survival and quality of life). The other is focused on how promising the response rate of a drug candidate have to be to be considered for oncology drug approval. People With More Brown Fat May Burn Fuel More Efficiently
 Dr. Keifer[/caption]
Florian Keifer, M.D., Ph.D
Associate Professor of Medicine
Division of Endocrinology and Metabolism
Department of Medicine III
Medical University of Vienna
MedicalResearch.com: What is the background for this study?
Response: Brown fat, in contrast to white fat, burns significant amounts of chemical energy through heat production. In numerous animal models the activation of brown adipose tissue (BAT) increases energy expenditure and counteracts weight gain. Therefore BAT has been established as a promising target in the fight against obesity and related metabolic disorders. In humans, BAT can be activated by moderate cold exposure, however the function and relevance of BAT are incompletely understood. Using PET scans we identified two groups of individuals those with and without active BAT and studied their differences in energy expenditure and blood fatty acid composition.
Dr. Keifer[/caption]
Florian Keifer, M.D., Ph.D
Associate Professor of Medicine
Division of Endocrinology and Metabolism
Department of Medicine III
Medical University of Vienna
MedicalResearch.com: What is the background for this study?
Response: Brown fat, in contrast to white fat, burns significant amounts of chemical energy through heat production. In numerous animal models the activation of brown adipose tissue (BAT) increases energy expenditure and counteracts weight gain. Therefore BAT has been established as a promising target in the fight against obesity and related metabolic disorders. In humans, BAT can be activated by moderate cold exposure, however the function and relevance of BAT are incompletely understood. Using PET scans we identified two groups of individuals those with and without active BAT and studied their differences in energy expenditure and blood fatty acid composition. Metastatic Breast Cancer: Tumor Profiling by Liquid Biopsy Can Help Drive Therapy
 Rebecca Nagy[/caption]
Rebecca Nagy
Vice President Medical Affairs
Guardant Health
MedicalResearch.com: What is the background for this study?
Response: Hormone receptor positive (HR+) breast cancer comprises roughly 75% of all cancers of the breast. While many of these cancers can be cured through multi-modality therapy, there remain many deaths due to metastatic spread to distant organs. These metastatic cancers are marked by their resilience in the face of potent targeted therapies and chemotherapies, with many tumors displaying an initial drug response followed by resistance.
Recently, genomic sequencing has identified recurrent, oncogenic alterations in HR+ metastatic breast cancer (MBC) with mutations in the catalytic alpha subunit of PI3K (PI3Kα, PIK3CA gene), in over 40% of cases. This has raised hopes for more durable disease control through precise inhibition of this driver oncogene.
The SOLAR-1 Phase III study of alpelisib combined with fulvestrant in PIK3CA-mutated HR+ MBC showed a markedly improved PFS over fulvestrant monotherapy but pervasive resistance nonetheless. To characterize the basis for such resistance to combination hormone plus PIK3CA targeted therapy, we conducted a detailed, longitudinal analysis of tumor and plasma circulating cell-freetumor DNA (ctDNA) among patients with HR+ MBC who participated in a phase I/II dose escalation study of alpelisib in combination with letrozole or exemestane.
Rebecca Nagy[/caption]
Rebecca Nagy
Vice President Medical Affairs
Guardant Health
MedicalResearch.com: What is the background for this study?
Response: Hormone receptor positive (HR+) breast cancer comprises roughly 75% of all cancers of the breast. While many of these cancers can be cured through multi-modality therapy, there remain many deaths due to metastatic spread to distant organs. These metastatic cancers are marked by their resilience in the face of potent targeted therapies and chemotherapies, with many tumors displaying an initial drug response followed by resistance.
Recently, genomic sequencing has identified recurrent, oncogenic alterations in HR+ metastatic breast cancer (MBC) with mutations in the catalytic alpha subunit of PI3K (PI3Kα, PIK3CA gene), in over 40% of cases. This has raised hopes for more durable disease control through precise inhibition of this driver oncogene.
The SOLAR-1 Phase III study of alpelisib combined with fulvestrant in PIK3CA-mutated HR+ MBC showed a markedly improved PFS over fulvestrant monotherapy but pervasive resistance nonetheless. To characterize the basis for such resistance to combination hormone plus PIK3CA targeted therapy, we conducted a detailed, longitudinal analysis of tumor and plasma circulating cell-freetumor DNA (ctDNA) among patients with HR+ MBC who participated in a phase I/II dose escalation study of alpelisib in combination with letrozole or exemestane. Asthma Exacerbation Severity Is Greater for Women than for Men
 Olga Ryan, DrPH, MPH, MBA
Regional Clinical Account Director, Southwest
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Asthma is common and imparts a substantial societal burden. It is well documented that asthma prevalence varies between males and females. Before puberty, more boys have asthma. Following puberty, a greater proportion of women suffer with asthma. We also have observed that women experience greater morbidity from the illness, greater healthcare resource utilization and suboptimal response for guideline recommended therapies (ICS, ICS/LABA).
Rationale for this study focused on describing asthma related outcomes between a well characterized severe asthma cohort, with intent in delineating differences among the sexes. With the advent of targeted biological medicines for severe asthma, as well as apparent gaps in knowledge, we wanted to understand potential sex-specific disease indicators in a well characterized severe asthma cohort.
Olga Ryan, DrPH, MPH, MBA
Regional Clinical Account Director, Southwest
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Asthma is common and imparts a substantial societal burden. It is well documented that asthma prevalence varies between males and females. Before puberty, more boys have asthma. Following puberty, a greater proportion of women suffer with asthma. We also have observed that women experience greater morbidity from the illness, greater healthcare resource utilization and suboptimal response for guideline recommended therapies (ICS, ICS/LABA).
Rationale for this study focused on describing asthma related outcomes between a well characterized severe asthma cohort, with intent in delineating differences among the sexes. With the advent of targeted biological medicines for severe asthma, as well as apparent gaps in knowledge, we wanted to understand potential sex-specific disease indicators in a well characterized severe asthma cohort.
Lifetime Health Care Costs Associated with Systemic Corticosteroid Use in Adults with Persistent Asthma
 Yen Chung, PharmD
Payer Evidence Director
US Medical Affairs, AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: Among patients with persistent asthma, use of systemic corticosteroids (SCS) is typically reserved for treatment of asthma exacerbations and as a supplemental maintenance therapy for patients whose disease remains uncontrolled with maximum maintenance controller therapies. However, SCS therapy comes with known risks for acute and chronic complications. It is well established that patients with severe asthma are responsible for a disproportionate amount of the economic burden of asthma; however, less clear is the extent to which systemic corticosteroids use and its consequences specifically contributes to the cost burden of asthma.
The purpose of this study was to use administrative claims to follow asthma patients with and without SCS treatment for up to 3 years and compare their complication rates, health care resource utilization, and costs.
Yen Chung, PharmD
Payer Evidence Director
US Medical Affairs, AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: Among patients with persistent asthma, use of systemic corticosteroids (SCS) is typically reserved for treatment of asthma exacerbations and as a supplemental maintenance therapy for patients whose disease remains uncontrolled with maximum maintenance controller therapies. However, SCS therapy comes with known risks for acute and chronic complications. It is well established that patients with severe asthma are responsible for a disproportionate amount of the economic burden of asthma; however, less clear is the extent to which systemic corticosteroids use and its consequences specifically contributes to the cost burden of asthma.
The purpose of this study was to use administrative claims to follow asthma patients with and without SCS treatment for up to 3 years and compare their complication rates, health care resource utilization, and costs. COVID-19: Which NYC Borough Has Highest Rate of Hospitalizations and Deaths?
 Dr. Wadhera[/caption]
Rishi Wadhera, MD, MPP, MPhil
Instructor in Medicine at Harvard Medical School
Cardiologist,Beth Israel Deaconess Medical Center
MedicalResearch.com: What is the background for this study?
Response: With more than a million confirmed cases of COVID-19 in the United States, there is growing concern that low-income communities and racial/ethnic minorities may be disproportionately shouldering the burden of the pandemic. New York City, which is comprised of 5 boroughs (Bronx, Brooklyn, Manhattan, Queens, and Staten Island) with unique demographic and socioeconomic characteristics, has emerged as the epicenter of the COVID-19 outbreak.
Dr. Wadhera[/caption]
Rishi Wadhera, MD, MPP, MPhil
Instructor in Medicine at Harvard Medical School
Cardiologist,Beth Israel Deaconess Medical Center
MedicalResearch.com: What is the background for this study?
Response: With more than a million confirmed cases of COVID-19 in the United States, there is growing concern that low-income communities and racial/ethnic minorities may be disproportionately shouldering the burden of the pandemic. New York City, which is comprised of 5 boroughs (Bronx, Brooklyn, Manhattan, Queens, and Staten Island) with unique demographic and socioeconomic characteristics, has emerged as the epicenter of the COVID-19 outbreak.Spinal Muscular Atrophy: Biogen Enrolls First Patient in Higher-Dose SPINRAZA® (NUSINERSEN) Study
 Dr. Ferguson[/caption]
Toby Ferguson, M.D., Ph.D.
Vice President, Head Neuromuscular Development Unit
Biogen
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by SMA, who is primarily affected and incidence?
Response: Spinal muscular atrophy (SMA) is a rare, genetic, neuromuscular disease characterized by a loss of motor neurons in the spinal cord and lower brain stem that can result in severe, progressive muscle atrophy and weakness. Approximately one in 10,000 live births have a diagnosis of SMA. It is a leading genetic cause of infant mortality; however, people of all ages are impacted by the disease.
More than three years ago, SPINRAZA (nusinersen) became the first FDA-approved treatment option for SMA.
The DEVOTE study, which recently treated its first patient, is designed to evaluate the safety and potential for even greater efficacy of SPINRAZA when administered at a higher dose than currently approved for the treatment of SMA. The Phase 2/3 randomized, controlled, dose-escalating study will be conducted at approximately 50 sites around the world and aims to enroll individuals of all ages with SMA.
Dr. Ferguson[/caption]
Toby Ferguson, M.D., Ph.D.
Vice President, Head Neuromuscular Development Unit
Biogen
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by SMA, who is primarily affected and incidence?
Response: Spinal muscular atrophy (SMA) is a rare, genetic, neuromuscular disease characterized by a loss of motor neurons in the spinal cord and lower brain stem that can result in severe, progressive muscle atrophy and weakness. Approximately one in 10,000 live births have a diagnosis of SMA. It is a leading genetic cause of infant mortality; however, people of all ages are impacted by the disease.
More than three years ago, SPINRAZA (nusinersen) became the first FDA-approved treatment option for SMA.
The DEVOTE study, which recently treated its first patient, is designed to evaluate the safety and potential for even greater efficacy of SPINRAZA when administered at a higher dose than currently approved for the treatment of SMA. The Phase 2/3 randomized, controlled, dose-escalating study will be conducted at approximately 50 sites around the world and aims to enroll individuals of all ages with SMA.
Over 80% of World Trade First Responders Have Fatty Liver Disease
 D. Debashis Reja[/caption]
Mishal Reja, MD, MD
Resident in Internal Medicine
Robert Wood Johnson University Hospital
MedicalResearch.com: What is the background for this study?
Response: World Trade Center first responders were exposed to environmental toxicants that have resulted in negative health consequences.
Gastrointestinal aerodigestive disorders such as GERD and Barrett’s esophagus have been frequently reported in this population. Additionally, an increasing body of literature has shown that fatty liver disease is not only secondary to metabolic syndrome, diabetes, and obesity, as previously thought, but can also result from environmental and industrial toxicants. Chemicals such as Vinyl Chloride, Tetrachloroethylene, Perchloroethylene, and many others are frequently found in industrial occupations and have resulted in fatty liver disease, steatohepatitis, cirrhosis, and hepatocellular carcinoma. Many of these chemicals were also present at ground zero, thus exposing many of first responders to the hepatotoxic effects of these chemicals. To date this is the first study to look at liver disease in World Trade Center first responders.
D. Debashis Reja[/caption]
Mishal Reja, MD, MD
Resident in Internal Medicine
Robert Wood Johnson University Hospital
MedicalResearch.com: What is the background for this study?
Response: World Trade Center first responders were exposed to environmental toxicants that have resulted in negative health consequences.
Gastrointestinal aerodigestive disorders such as GERD and Barrett’s esophagus have been frequently reported in this population. Additionally, an increasing body of literature has shown that fatty liver disease is not only secondary to metabolic syndrome, diabetes, and obesity, as previously thought, but can also result from environmental and industrial toxicants. Chemicals such as Vinyl Chloride, Tetrachloroethylene, Perchloroethylene, and many others are frequently found in industrial occupations and have resulted in fatty liver disease, steatohepatitis, cirrhosis, and hepatocellular carcinoma. Many of these chemicals were also present at ground zero, thus exposing many of first responders to the hepatotoxic effects of these chemicals. To date this is the first study to look at liver disease in World Trade Center first responders.
Fecal Microbiota Transplant For Multi-Drug-Resistant Organisms: Improved clinical Outcomes Beyond Intestinal Decolonization
 Prof. Marchesi[/caption]
Prof. Julian Marchesi PhD
Professor of Digestive Health
Faculty of Medicine, Department of Metabolism, Digestion and Reproduction
[caption id="attachment_54021" align="alignleft" width="149"]
Prof. Marchesi[/caption]
Prof. Julian Marchesi PhD
Professor of Digestive Health
Faculty of Medicine, Department of Metabolism, Digestion and Reproduction
[caption id="attachment_54021" align="alignleft" width="149"] Prof. Mullish[/caption]
Dr. Benjamin Mullish PhD
Faculty of Medicine, Department of Metabolism, Digestion and Reproduction
NIHR Clinical Lecturer
Imperial College London
MedicalResearch.com: What is the background for this study?
Response: Many patients are colonized with bacteria that are resistant to nearly all the antibiotics that we currently have. This antibiotic resistance is a huge public health problem, not least because it may lead to the scenario where a bacterial species moves from the gut and into the bloodstream, causes an infection, and cannot be treated. Such scenarios particularly occur in patients who are particularly prone to getting multiple and frequent courses of antibiotics; this may include patients with particular kidney conditions (who may be vulnerable to recurrent urinary tract infections (UTIs)), and patients with blood cancers (such as leukaemia, who have weak immune systems and are therefore prone to infections).
Furthermore, in both sets of patients, to help treat their disease, they may be offered transplants, either a new kidney or new bone marrow. When this transplant happens, the clinician needs to ‘switch off’ their immune system to allow the transplant to work. When the immune system is dialled down, it can no longer stop any invading bacteria, increasing the chance of antibiotic resistance bacteria causing infections, which frequently leads to patient death.
Prof. Mullish[/caption]
Dr. Benjamin Mullish PhD
Faculty of Medicine, Department of Metabolism, Digestion and Reproduction
NIHR Clinical Lecturer
Imperial College London
MedicalResearch.com: What is the background for this study?
Response: Many patients are colonized with bacteria that are resistant to nearly all the antibiotics that we currently have. This antibiotic resistance is a huge public health problem, not least because it may lead to the scenario where a bacterial species moves from the gut and into the bloodstream, causes an infection, and cannot be treated. Such scenarios particularly occur in patients who are particularly prone to getting multiple and frequent courses of antibiotics; this may include patients with particular kidney conditions (who may be vulnerable to recurrent urinary tract infections (UTIs)), and patients with blood cancers (such as leukaemia, who have weak immune systems and are therefore prone to infections).
Furthermore, in both sets of patients, to help treat their disease, they may be offered transplants, either a new kidney or new bone marrow. When this transplant happens, the clinician needs to ‘switch off’ their immune system to allow the transplant to work. When the immune system is dialled down, it can no longer stop any invading bacteria, increasing the chance of antibiotic resistance bacteria causing infections, which frequently leads to patient death. New Gene Regions Linked to Melanoma Risk Identified
 Dr. Law[/caption]
Dr. Matthew H. Law, PhD
Senior Research Officer, Statistical Genetics
QIMR Berghofer
MedicalResearch.com: What is the background for this study?
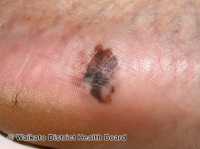
Response: A large genetic study of melanoma involving a global collaboration of scientists, co-led by QIMR Berghofer, the University of Leeds in the UK, and the National Cancer Institute in the US which is part of the National Institutes of Health, has been published in the prestigious journal Nature Genetics.
Melanoma is a sometimes-deadly skin cancer, with an estimated 350,000 cases worldwide in 2015, resulting in nearly 60,000 deaths. Melanoma begins in melanocytes, cells in the skin responsible for making the pigment melanin that gives colour to the skin. Melanin is able to block some of the harmful effects of UV radiation, which is why people with pale skin are at a higher risk of skin cancer, but the protection is not complete. Moles also develop from melanocytes, and having a high number of moles is a risk factor for melanoma.
UK based co-lead author, Dr Mark Iles from the University of Leeds’s Institute for Data Analytics, said the researchers examined DNA from 37,000 people who had been diagnosed with melanoma and compared their genetic information to that of nearly 400,000 people with no history of the disease.”
Joint study leader and QIMR Berghofer statistical geneticist Associate Professor Matthew Law said the researchers identified 33 new regions of the genome and confirmed another 21 previously reported regions that are linked to a person’s risk of developing melanoma of the skin. Two of the new regions we’ve discovered that are linked to melanoma have previously been linked to autoimmune disorders. This provides further evidence that the immune system plays an important role in a person developing melanoma. We also found an association between melanoma and common genetic variants in the gene TP53, which is a gene critical in controlling DNA repair when cells divide, and in suppressing cancer.”
Co-lead author on the study and senior investigator at the National Cancer Institute, Dr Maria Teresa Landi, said the research also uncovered other important clues to the genetic causes of melanoma. We used the relationship between moles, pigmentation, and melanoma to identify 31 additional gene regions that potentially influence melanoma risk. For example, one of the regions we identified is involved in melanocyte growth,” Dr Landi said. “Moreover, we also included people from Mediterranean populations involved in the MelaNostrum Consortium. Most studies of melanoma use people with northern or western European ancestry (e.g. British) and by expanding our analysis to include Mediterranean populations, we will gain a greater understanding of the genetics of melanoma in this highly sun exposed group.”
Dr. Law[/caption]
Dr. Matthew H. Law, PhD
Senior Research Officer, Statistical Genetics
QIMR Berghofer
MedicalResearch.com: What is the background for this study?
Response: A large genetic study of melanoma involving a global collaboration of scientists, co-led by QIMR Berghofer, the University of Leeds in the UK, and the National Cancer Institute in the US which is part of the National Institutes of Health, has been published in the prestigious journal Nature Genetics.
Melanoma is a sometimes-deadly skin cancer, with an estimated 350,000 cases worldwide in 2015, resulting in nearly 60,000 deaths. Melanoma begins in melanocytes, cells in the skin responsible for making the pigment melanin that gives colour to the skin. Melanin is able to block some of the harmful effects of UV radiation, which is why people with pale skin are at a higher risk of skin cancer, but the protection is not complete. Moles also develop from melanocytes, and having a high number of moles is a risk factor for melanoma.
UK based co-lead author, Dr Mark Iles from the University of Leeds’s Institute for Data Analytics, said the researchers examined DNA from 37,000 people who had been diagnosed with melanoma and compared their genetic information to that of nearly 400,000 people with no history of the disease.”
Joint study leader and QIMR Berghofer statistical geneticist Associate Professor Matthew Law said the researchers identified 33 new regions of the genome and confirmed another 21 previously reported regions that are linked to a person’s risk of developing melanoma of the skin. Two of the new regions we’ve discovered that are linked to melanoma have previously been linked to autoimmune disorders. This provides further evidence that the immune system plays an important role in a person developing melanoma. We also found an association between melanoma and common genetic variants in the gene TP53, which is a gene critical in controlling DNA repair when cells divide, and in suppressing cancer.”
Co-lead author on the study and senior investigator at the National Cancer Institute, Dr Maria Teresa Landi, said the research also uncovered other important clues to the genetic causes of melanoma. We used the relationship between moles, pigmentation, and melanoma to identify 31 additional gene regions that potentially influence melanoma risk. For example, one of the regions we identified is involved in melanocyte growth,” Dr Landi said. “Moreover, we also included people from Mediterranean populations involved in the MelaNostrum Consortium. Most studies of melanoma use people with northern or western European ancestry (e.g. British) and by expanding our analysis to include Mediterranean populations, we will gain a greater understanding of the genetics of melanoma in this highly sun exposed group.”Eczema and Psoriasis: Itch Linked to Specific Protein in Skin
 Dr. Mishra[/caption]
Santosh K. Mishra M.Tech., PhD
Assistant Professor of Neuroscience
Department of Molecular Biomedical Sciences
NC State Veterinary Medicine
Raleigh, NC 2760
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by atopic dermatitis?
Response: Chronic allergic itch is a worldwide problem that leads to substantial health expenses,but what causes this universal urge to scratch remains elusive in chronic allergic itch. Atopic dermatitis is a common allergic skin disease that often associated with extremely itchy and inflamed skin.
In our study, we showed, for the first time, a molecular pathway that is involved in chronic allergic itch as we identified an endogenous mediator (periostin) and a new role for its sensory neuron receptor, the integrin αVβ3, which drives the excitability and transmission of itch signal to the spinal cord.
Dr. Mishra[/caption]
Santosh K. Mishra M.Tech., PhD
Assistant Professor of Neuroscience
Department of Molecular Biomedical Sciences
NC State Veterinary Medicine
Raleigh, NC 2760
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by atopic dermatitis?
Response: Chronic allergic itch is a worldwide problem that leads to substantial health expenses,but what causes this universal urge to scratch remains elusive in chronic allergic itch. Atopic dermatitis is a common allergic skin disease that often associated with extremely itchy and inflamed skin.
In our study, we showed, for the first time, a molecular pathway that is involved in chronic allergic itch as we identified an endogenous mediator (periostin) and a new role for its sensory neuron receptor, the integrin αVβ3, which drives the excitability and transmission of itch signal to the spinal cord. Widespread Public Support and Compliance for Stay-at-Home Policies
 Mark Czeisler[/caption]
Mark Czeisler MPhil
-2020 Fulbright Future Scholar
Australian-American Fulbright Commission
Funded by the Kinghorn Family Foundation
-Honorary Research Fellow, Institute for Breathing and Sleep Austin Health
-Master’s candidate School of Psychological Sciences and
Turner Institute for Brain and Mental Health
Monash University
MedicalResearch.com: What is the background for this study?
Response: Recognizing the difficult decisions of when and how to manage stringent COVID-19 mitigation strategies faced by health officials and policymakers, researchers at Brigham and Women’s Hospital and Monash University sought to assess public compliance with and support for the current mitigation strategies (e.g., quarantine, stay-at-home orders). We also assessed the life impact of such stringent tactics.
We acquired nationally demographically representative samples from one nation and city with large numbers of COVID-19 infections and deaths (US and New York City) and one nation and city with comparatively small numbers of COVID-19 infections and deaths (Australia and Los Angeles).
Mark Czeisler[/caption]
Mark Czeisler MPhil
-2020 Fulbright Future Scholar
Australian-American Fulbright Commission
Funded by the Kinghorn Family Foundation
-Honorary Research Fellow, Institute for Breathing and Sleep Austin Health
-Master’s candidate School of Psychological Sciences and
Turner Institute for Brain and Mental Health
Monash University
MedicalResearch.com: What is the background for this study?
Response: Recognizing the difficult decisions of when and how to manage stringent COVID-19 mitigation strategies faced by health officials and policymakers, researchers at Brigham and Women’s Hospital and Monash University sought to assess public compliance with and support for the current mitigation strategies (e.g., quarantine, stay-at-home orders). We also assessed the life impact of such stringent tactics.
We acquired nationally demographically representative samples from one nation and city with large numbers of COVID-19 infections and deaths (US and New York City) and one nation and city with comparatively small numbers of COVID-19 infections and deaths (Australia and Los Angeles).
CRISPR-Edited Stem Cells Cured Diabetes in a Mouse Model
 Dr. Urano[/caption]
Fumihiko Urano, MD, PhD
Samuel E. Schechter Professor of Medicine
Division of Endocrinology, Metabolism, and Lipid Research
Washington University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Wolfram syndrome is a rare monogenic disease characterized by insulin-dependent diabetes, retinal degeneration, and neurodegeneration. Using gene editing by CRISPR-Cas9, in combination with patient-derived induced pluripotent stem cells (iPSCs), we were able to make normal insulin-producing pancreatic beta cells by correcting Wolfram Syndrome gene mutation.
We could cure diabetes in cells and mice. Because we can create any types of tissues from iPSCs, our next step would be to replicate this success for other medical problems, including retinal regeneration and neurodegeneration.
Dr. Urano[/caption]
Fumihiko Urano, MD, PhD
Samuel E. Schechter Professor of Medicine
Division of Endocrinology, Metabolism, and Lipid Research
Washington University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Wolfram syndrome is a rare monogenic disease characterized by insulin-dependent diabetes, retinal degeneration, and neurodegeneration. Using gene editing by CRISPR-Cas9, in combination with patient-derived induced pluripotent stem cells (iPSCs), we were able to make normal insulin-producing pancreatic beta cells by correcting Wolfram Syndrome gene mutation.
We could cure diabetes in cells and mice. Because we can create any types of tissues from iPSCs, our next step would be to replicate this success for other medical problems, including retinal regeneration and neurodegeneration.RECOVER Study: Recovery From Opioid Use Disorder After Monthly Long-Acting Buprenorphine Treatment
 Dr. Ling[/caption]
Dr. Walter Ling, MD
Professor of Psychiatry
Director of Integrated Substance Abuse Programs UCLA
MedicalResearch.com: What is the background for this study?
Response: RECOVER™ is a real-world, observational study looking at long-term recovery in a cohort of 533 people with moderate to severe opioid use disorder (OUD) following their transition from two Phase 3 clinical trials of SUBLOCADE® (buprenorphine extended-release) injection, for subcutaneous use (CIII), into a real-world setting.1
The RECOVER study uses data from three main sources: self-administered assessments from enrolled individuals, urine drug screens (UDS) and data collected from several public sources. Recovery is examined over 24 months – the self-administered assessment and UDS results are completed by participants every three months over the course of this period. Results are being analyzed to understand the clinical, socio-economic and environmental factors associated with continuous effects of medications to treat OUD after a clinical trial.1.2
Studies such as RECOVER can help bridge the knowledge gap between the efficacy of medications as seen in the controlled clinical trial environment, and the use and effect of medications outside of a research setting and their long-term impact on patients’ health. A collaboration between Indivior and the Fralin Biomedical Institute at Virginia Tech Carilion will enable the next phase of the RECOVER study, which may provide further information to health care providers and policymakers on how to use medications to support their patients and how continuity of care can help break down barriers to evidence-based treatment.3
Dr. Ling[/caption]
Dr. Walter Ling, MD
Professor of Psychiatry
Director of Integrated Substance Abuse Programs UCLA
MedicalResearch.com: What is the background for this study?
Response: RECOVER™ is a real-world, observational study looking at long-term recovery in a cohort of 533 people with moderate to severe opioid use disorder (OUD) following their transition from two Phase 3 clinical trials of SUBLOCADE® (buprenorphine extended-release) injection, for subcutaneous use (CIII), into a real-world setting.1
The RECOVER study uses data from three main sources: self-administered assessments from enrolled individuals, urine drug screens (UDS) and data collected from several public sources. Recovery is examined over 24 months – the self-administered assessment and UDS results are completed by participants every three months over the course of this period. Results are being analyzed to understand the clinical, socio-economic and environmental factors associated with continuous effects of medications to treat OUD after a clinical trial.1.2
Studies such as RECOVER can help bridge the knowledge gap between the efficacy of medications as seen in the controlled clinical trial environment, and the use and effect of medications outside of a research setting and their long-term impact on patients’ health. A collaboration between Indivior and the Fralin Biomedical Institute at Virginia Tech Carilion will enable the next phase of the RECOVER study, which may provide further information to health care providers and policymakers on how to use medications to support their patients and how continuity of care can help break down barriers to evidence-based treatment.3 Possible Mechanisms Linking Obesity and COVID-19 Severity
 Prof. Stefan[/caption]
Univ.-Prof. Norbert Stefan, MD
-Heisenberg Professorship for
Clinical and Experimental Diabetology
Internal Medicine IV, University of Tübingen
-Head of the Department of Pathophysiology of
Prediabetes at the Institute of Diabetes Research and
Metabolic Diseases (IDM) of the Helmholtz Center Munich
-Visiting Professor Harvard Medical School, Boston, USA
MedicalResearch.com: What is the background for this study?
Response: Working in the field of obesity and cardiometabolic diseases my colleagues and I were very surprised that most of the articles reporting data about comorbid conditions, which may be associated with increased risk of severe COVID-19, did not provide data about body fat mass.
Because increased fat mass, and more so higher upper-body fat mass, are known to strongly predict an increased risk of cardiometabolic disease, pneumonia and mortality, we hypothesized that they may also predict a more severe course of COVID-19.
Prof. Stefan[/caption]
Univ.-Prof. Norbert Stefan, MD
-Heisenberg Professorship for
Clinical and Experimental Diabetology
Internal Medicine IV, University of Tübingen
-Head of the Department of Pathophysiology of
Prediabetes at the Institute of Diabetes Research and
Metabolic Diseases (IDM) of the Helmholtz Center Munich
-Visiting Professor Harvard Medical School, Boston, USA
MedicalResearch.com: What is the background for this study?
Response: Working in the field of obesity and cardiometabolic diseases my colleagues and I were very surprised that most of the articles reporting data about comorbid conditions, which may be associated with increased risk of severe COVID-19, did not provide data about body fat mass.
Because increased fat mass, and more so higher upper-body fat mass, are known to strongly predict an increased risk of cardiometabolic disease, pneumonia and mortality, we hypothesized that they may also predict a more severe course of COVID-19.
Limiting the Spread: 6 Core Infection Control Principles That Apply to any Healthcare Environment
It only takes a second for an infection to start. That's why medical professionals must use standard precautions to prevent...
Opioid Use Disorder Drugs: Removing Need for Prior Authorization Can Save Money in the Long Run
 Dr. Mark[/caption]
Tami L. Mark, PhD
Senior Director, Behavioral Health Financing and Quality Measurement
RTI International
MedicalResearch.com: What is the background for this study?
Response: There are effective medications to treat opioid use disorder. Federal and state policymakers have tried to improve access to these medications. However, medications to treat opioid use disorders are still often subject to prior authorization. Studies of other medications finds that prior authorization can reduce access.
This study looked at whether removing prior authorization in Medicare Part D plans was associated with increases in the use of medications to treat opioid use disorder.
Dr. Mark[/caption]
Tami L. Mark, PhD
Senior Director, Behavioral Health Financing and Quality Measurement
RTI International
MedicalResearch.com: What is the background for this study?
Response: There are effective medications to treat opioid use disorder. Federal and state policymakers have tried to improve access to these medications. However, medications to treat opioid use disorders are still often subject to prior authorization. Studies of other medications finds that prior authorization can reduce access.
This study looked at whether removing prior authorization in Medicare Part D plans was associated with increases in the use of medications to treat opioid use disorder. Adding a Filtered Eye Mask May Reduce COVID-19 Infections
 Dr. Douglas[/caption]
David B. Douglas, M.D., M.P.H.
Harvard T.H. Chan School of Public Health
MedicalResearch.com: What is the background for this study?
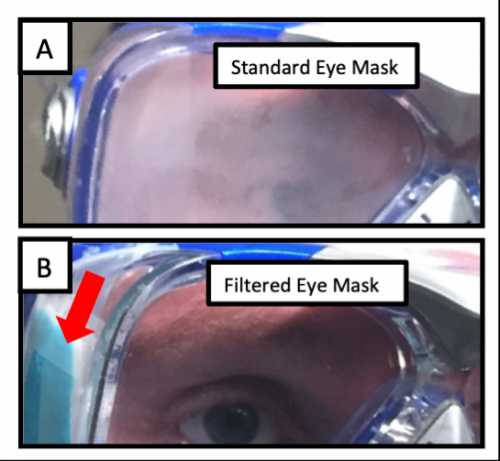
Response: Health care workers are facing the major threat of catching COVID-19 through their eyes, yet currently available eye protection is inadequate. Specifically, the use of open-type eye protection allows airborne viruses to float over the top, around the sides or under the bottom of the lenses and contact the eyes. Additionally, eye protection is well known to fog up, which limits usability by making even the most basic tasks challenging. In fact, fogged goggles is a major barrier to use.
[caption id="attachment_53981" align="aligncenter" width="500"]
Dr. Douglas[/caption]
David B. Douglas, M.D., M.P.H.
Harvard T.H. Chan School of Public Health
MedicalResearch.com: What is the background for this study?
Response: Health care workers are facing the major threat of catching COVID-19 through their eyes, yet currently available eye protection is inadequate. Specifically, the use of open-type eye protection allows airborne viruses to float over the top, around the sides or under the bottom of the lenses and contact the eyes. Additionally, eye protection is well known to fog up, which limits usability by making even the most basic tasks challenging. In fact, fogged goggles is a major barrier to use.
[caption id="attachment_53981" align="aligncenter" width="500"]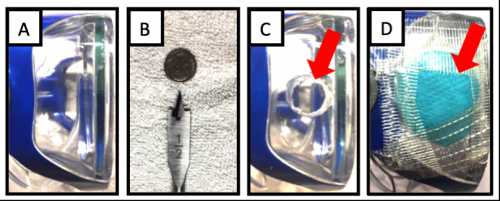 The building of the filtered eye mask prototype.[/caption]
The building of the filtered eye mask prototype.[/caption]
EVALI: E-Cigarette & Vaping Lung Injury Hospitalizations and Deaths
 Dr. Werner[/caption]
Angela K. Werner, PhD, MPH
Environmental Public Health Tracking Program
National Center for Environmental Health
CDC
MedicalResearch.com: What is the background for this study?
Response: In August 2019, emergency department (ED) visits related to e-cigarette, or vaping, product-associated lung injury (EVALI) increased sharply, followed by a peak in September. This was followed by a gradual but persistent decline in the number of cases.
As of February 18, 2020 (CDC’s latest and final published update), there were a total of 2,807 hospitalized EVALI cases or deaths reported to CDC from all 50 states, the District of Columbia, and two U.S. territories (Puerto Rico and U.S. Virgin Islands). Sixty-eight deaths were confirmed in 29 states and the District of Columbia. Although clinical presentations and outcomes of EVALI patients have been reported, data on patients who died are more limited. This article fills a gap by reporting on a national study assessing detailed demographic, substance use, and clinical characteristics of EVALI patients who have died, and comparing them to the characteristics of EVALI patients who survived.
Dr. Werner[/caption]
Angela K. Werner, PhD, MPH
Environmental Public Health Tracking Program
National Center for Environmental Health
CDC
MedicalResearch.com: What is the background for this study?
Response: In August 2019, emergency department (ED) visits related to e-cigarette, or vaping, product-associated lung injury (EVALI) increased sharply, followed by a peak in September. This was followed by a gradual but persistent decline in the number of cases.
As of February 18, 2020 (CDC’s latest and final published update), there were a total of 2,807 hospitalized EVALI cases or deaths reported to CDC from all 50 states, the District of Columbia, and two U.S. territories (Puerto Rico and U.S. Virgin Islands). Sixty-eight deaths were confirmed in 29 states and the District of Columbia. Although clinical presentations and outcomes of EVALI patients have been reported, data on patients who died are more limited. This article fills a gap by reporting on a national study assessing detailed demographic, substance use, and clinical characteristics of EVALI patients who have died, and comparing them to the characteristics of EVALI patients who survived.New Protocol for Rapid Exclusion of Heart Attack in a US Safety Net Hospital
 Dr. Vigen[/caption]
Rebecca Vigen, MD, MSCS
Assistant Professor of Internal Medicine
UT Southwestern
MedicalResearch.com: What is the background for this study?
Response: Emergency department overcrowding is an urgent health priority and chest pain is a common reason for emergency department visits. We developed a new protocol that uses high sensitivity cardiac troponin testing with a risk assessment tool that guides decisions on discharge and stress testing for patients presenting with chest pain. The protocol allows us to rule out heart attacks more quickly than the protocols utilizing an older troponin assay.
Dr. Vigen[/caption]
Rebecca Vigen, MD, MSCS
Assistant Professor of Internal Medicine
UT Southwestern
MedicalResearch.com: What is the background for this study?
Response: Emergency department overcrowding is an urgent health priority and chest pain is a common reason for emergency department visits. We developed a new protocol that uses high sensitivity cardiac troponin testing with a risk assessment tool that guides decisions on discharge and stress testing for patients presenting with chest pain. The protocol allows us to rule out heart attacks more quickly than the protocols utilizing an older troponin assay.Fewer EVALI Cases in States with Recreational Marijuana
 Dr. Hollingsworth[/caption]
Alex Hollingsworth PhD
Assistant Professor
O'Neill School of Public and Environmental Affairs
Indiana University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: I've been working with Coady Wing and Ashley Bradford on a few different studies of the effects of recreational marijuana laws on drug and alcohol use.
Soon after EVALI became a major issue, the prevailing theory from the CDC and others was that EVALI was caused by the use of vitamin E acetate in illegal THC vaping products.
Our group read about this and we thought about some of the things that often happen in black markets for illegal drugs. For instance, during the alcohol prohibition era, bootleg alcohol producers often made and sold alcohol products that were not that safe to drink. In more recent years, there are cases where black market sellers of illegal drugs like heroin try to increase profit margins by adding other substances, which can be harmful.
We thought that maybe something like that could be happening in EVALI. Perhaps people in states where recreational marijuana is legal tended to purchase marijuana products from the legal market and the legal market was not selling any marijuana vaping products that included vitamin E acetate.
Dr. Hollingsworth[/caption]
Alex Hollingsworth PhD
Assistant Professor
O'Neill School of Public and Environmental Affairs
Indiana University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: I've been working with Coady Wing and Ashley Bradford on a few different studies of the effects of recreational marijuana laws on drug and alcohol use.
Soon after EVALI became a major issue, the prevailing theory from the CDC and others was that EVALI was caused by the use of vitamin E acetate in illegal THC vaping products.
Our group read about this and we thought about some of the things that often happen in black markets for illegal drugs. For instance, during the alcohol prohibition era, bootleg alcohol producers often made and sold alcohol products that were not that safe to drink. In more recent years, there are cases where black market sellers of illegal drugs like heroin try to increase profit margins by adding other substances, which can be harmful.
We thought that maybe something like that could be happening in EVALI. Perhaps people in states where recreational marijuana is legal tended to purchase marijuana products from the legal market and the legal market was not selling any marijuana vaping products that included vitamin E acetate.COVID-19: Low Sensitivity of Rapid Point-of-Care Testing in a Community Setting
 Dr. Döhla[/caption]
Dr. med. Manuel Döhla
Associate Researcher, AG "One Health"
Institute for Hygiene and Public Health
Medical Faculty, University of Bonn
Bonn, Germany
MedicalResearch.com: What is the background for this study?
Response: Rapid and reliable testing of suspected cases is an important measure in the fight against the corona pandemic. In PCR diagnostics, 24 hours and sometimes more elapse between testing and notification of the test result. During this period, suspect cases must be isolated pre-emptively so that they do not cause further transmission. This is logistically and personnel-intensive and highly error-prone.
The shorter the time between test and result, the more effective containment measures can be. This is why the test we have evaluated, which shows a result within 20 minutes, has aroused our interest. The manufacturer's specifications (sensitivity 70 % in early stage disease (day 4-10), 100 % in late stage disease (day 11-24), specificity 100 %) were promising.
Dr. Döhla[/caption]
Dr. med. Manuel Döhla
Associate Researcher, AG "One Health"
Institute for Hygiene and Public Health
Medical Faculty, University of Bonn
Bonn, Germany
MedicalResearch.com: What is the background for this study?
Response: Rapid and reliable testing of suspected cases is an important measure in the fight against the corona pandemic. In PCR diagnostics, 24 hours and sometimes more elapse between testing and notification of the test result. During this period, suspect cases must be isolated pre-emptively so that they do not cause further transmission. This is logistically and personnel-intensive and highly error-prone.
The shorter the time between test and result, the more effective containment measures can be. This is why the test we have evaluated, which shows a result within 20 minutes, has aroused our interest. The manufacturer's specifications (sensitivity 70 % in early stage disease (day 4-10), 100 % in late stage disease (day 11-24), specificity 100 %) were promising.Effect of Mediterranean Diet on Cognitive Function in Patients with Age-Related Eye Disease
 Dr. Chew[/caption]
Emily Y. Chew, M.D.
Director of the Division of Epidemiology and Clinical Applications
Deputy Clinical Director at the National Eye Institute (NEI),
National Institutes of Health
MedicalResearch.com: What is the background for this study?
Response: Dementia is a common disorder that was estimated to have a worldwide prevalence of 44 million in 2016 and is projected to hit 115 million by 2050. Many phase 3 trials of various therapies have failed and we have no treatment currently available for the prevention or reduction of the course of dementia. A slow neurocognitive decline throughout life is part of the normal process of aging. However, there is a subset of individuals who may have accelerated aging and is at high risk of development dementia. If the course of such accelerated decline could be altered in any way, it would be important to evaluate. The role of diet with biologic aging has been studied and diet has been also found to be associated with age-related conditions linked to dementia, including cardiovascular disease and diabetes.
We were interested in the cognitive function of our participants who had another neurodegenerative disease, age-related macular degeneration (AMD). We had conducted two randomized controlled clinical trials designed to evaluate the role of oral supplements for the treatment of AMD. We also studied cognitive function in both clinical trials of nearly 8,000 participants who were followed for 10 years. We also evaluated the dietary habits of the participants with food frequency questionnaires (FFQ) at baseline. Cognitive function testing was conducted in the first study, the Age-Related Eye Disease Study (AREDS) near the end of the clinical trial while the AREDS2, the second study, evaluated cognitive function testing at baseline and every 2 years until year 6. AREDS study evaluated cognitive function with in-clinic study visits while AREDS2 was conducted using telephone interviews.
Our aim was to determine whether closer adherence to the alternative Mediterranean diet (aMED) was associated with impaired cognitive function these two studies. We were interested in the particular components of the Mediterranean diet that may be important. We also evaluated the interaction of genetics with the diet.
Dr. Chew[/caption]
Emily Y. Chew, M.D.
Director of the Division of Epidemiology and Clinical Applications
Deputy Clinical Director at the National Eye Institute (NEI),
National Institutes of Health
MedicalResearch.com: What is the background for this study?
Response: Dementia is a common disorder that was estimated to have a worldwide prevalence of 44 million in 2016 and is projected to hit 115 million by 2050. Many phase 3 trials of various therapies have failed and we have no treatment currently available for the prevention or reduction of the course of dementia. A slow neurocognitive decline throughout life is part of the normal process of aging. However, there is a subset of individuals who may have accelerated aging and is at high risk of development dementia. If the course of such accelerated decline could be altered in any way, it would be important to evaluate. The role of diet with biologic aging has been studied and diet has been also found to be associated with age-related conditions linked to dementia, including cardiovascular disease and diabetes.
We were interested in the cognitive function of our participants who had another neurodegenerative disease, age-related macular degeneration (AMD). We had conducted two randomized controlled clinical trials designed to evaluate the role of oral supplements for the treatment of AMD. We also studied cognitive function in both clinical trials of nearly 8,000 participants who were followed for 10 years. We also evaluated the dietary habits of the participants with food frequency questionnaires (FFQ) at baseline. Cognitive function testing was conducted in the first study, the Age-Related Eye Disease Study (AREDS) near the end of the clinical trial while the AREDS2, the second study, evaluated cognitive function testing at baseline and every 2 years until year 6. AREDS study evaluated cognitive function with in-clinic study visits while AREDS2 was conducted using telephone interviews.
Our aim was to determine whether closer adherence to the alternative Mediterranean diet (aMED) was associated with impaired cognitive function these two studies. We were interested in the particular components of the Mediterranean diet that may be important. We also evaluated the interaction of genetics with the diet. Increased Mortality Among Children in Foster Care
 Barbara Chaiyachati, MD PhD
SafePlace: The Center for Child Protection and Health
Division of General Pediatrics
The Children’s Hospital of Philadelphia,
Center for Pediatric Clinical Effectiveness
University of Pennsylvania
Philadelphia, Pennsylvania
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Children in foster care have high rates of medical problems including chronic diseases. There is less known about the differences in mortality for children in foster care.
Looking at national data from 2003 to 2016, this study finds that children (ages 1 to 18) in foster care have higher mortality compared to children in the general population and that the difference in mortality has increased over time.
Barbara Chaiyachati, MD PhD
SafePlace: The Center for Child Protection and Health
Division of General Pediatrics
The Children’s Hospital of Philadelphia,
Center for Pediatric Clinical Effectiveness
University of Pennsylvania
Philadelphia, Pennsylvania
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Children in foster care have high rates of medical problems including chronic diseases. There is less known about the differences in mortality for children in foster care.
Looking at national data from 2003 to 2016, this study finds that children (ages 1 to 18) in foster care have higher mortality compared to children in the general population and that the difference in mortality has increased over time.