Author Interviews / 18.03.2019
Knee Replacement: Benefits and Risks of Antibiotic-Loaded Bone Cement
MedicalResearch.com Interview with:
[caption id="attachment_47740" align="alignleft" width="130"] Dr. Chen[/caption]
Darwin Chen, MD
Assistant Professor of Orthopedic Surgery
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Periprosthetic joint infection unfortunately remains a leading cause of total knee arthroplasty failure. One method of mitigating the risk of PJI is to use antibiotic loaded bone cement in a prophylactic fashion.
While the use of antibiotic cement makes inherent sense, the decision is not as simple as it seems. There are potential side effects such as renal damage, antibiotic hypersensitivity, and antibiotic resistance. Antibiotics decrease the mechanical strength of cement fixation, which may impact component loosening. Additionally, antibiotic cement is significantly more expensive than standard cement, driving up cost. Currently there is no consensus on if antibiotic cement truly reduces infection risk and there are many conflicting studies.
The purpose of our study is the use a large national database to evaluate real world utilization patterns of antibiotic cement, and assess outcomes, complications, and cost associated with antibiotic cement usage. Our hypothesis was that antibiotic cement is associated with a decreased risk of infection and no increased risk of systemic complications.
Dr. Chen[/caption]
Darwin Chen, MD
Assistant Professor of Orthopedic Surgery
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Periprosthetic joint infection unfortunately remains a leading cause of total knee arthroplasty failure. One method of mitigating the risk of PJI is to use antibiotic loaded bone cement in a prophylactic fashion.
While the use of antibiotic cement makes inherent sense, the decision is not as simple as it seems. There are potential side effects such as renal damage, antibiotic hypersensitivity, and antibiotic resistance. Antibiotics decrease the mechanical strength of cement fixation, which may impact component loosening. Additionally, antibiotic cement is significantly more expensive than standard cement, driving up cost. Currently there is no consensus on if antibiotic cement truly reduces infection risk and there are many conflicting studies.
The purpose of our study is the use a large national database to evaluate real world utilization patterns of antibiotic cement, and assess outcomes, complications, and cost associated with antibiotic cement usage. Our hypothesis was that antibiotic cement is associated with a decreased risk of infection and no increased risk of systemic complications.
 Dr. Chen[/caption]
Darwin Chen, MD
Assistant Professor of Orthopedic Surgery
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Periprosthetic joint infection unfortunately remains a leading cause of total knee arthroplasty failure. One method of mitigating the risk of PJI is to use antibiotic loaded bone cement in a prophylactic fashion.
While the use of antibiotic cement makes inherent sense, the decision is not as simple as it seems. There are potential side effects such as renal damage, antibiotic hypersensitivity, and antibiotic resistance. Antibiotics decrease the mechanical strength of cement fixation, which may impact component loosening. Additionally, antibiotic cement is significantly more expensive than standard cement, driving up cost. Currently there is no consensus on if antibiotic cement truly reduces infection risk and there are many conflicting studies.
The purpose of our study is the use a large national database to evaluate real world utilization patterns of antibiotic cement, and assess outcomes, complications, and cost associated with antibiotic cement usage. Our hypothesis was that antibiotic cement is associated with a decreased risk of infection and no increased risk of systemic complications.
Dr. Chen[/caption]
Darwin Chen, MD
Assistant Professor of Orthopedic Surgery
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Periprosthetic joint infection unfortunately remains a leading cause of total knee arthroplasty failure. One method of mitigating the risk of PJI is to use antibiotic loaded bone cement in a prophylactic fashion.
While the use of antibiotic cement makes inherent sense, the decision is not as simple as it seems. There are potential side effects such as renal damage, antibiotic hypersensitivity, and antibiotic resistance. Antibiotics decrease the mechanical strength of cement fixation, which may impact component loosening. Additionally, antibiotic cement is significantly more expensive than standard cement, driving up cost. Currently there is no consensus on if antibiotic cement truly reduces infection risk and there are many conflicting studies.
The purpose of our study is the use a large national database to evaluate real world utilization patterns of antibiotic cement, and assess outcomes, complications, and cost associated with antibiotic cement usage. Our hypothesis was that antibiotic cement is associated with a decreased risk of infection and no increased risk of systemic complications.



 Dr. Rattray[/caption]
Nicholas A. Rattray, Ph.D.
Research Scientist/Investigator
VA HSR&D Center for Health Information and Communication
Implementation Core, Precision Monitoring to Transform Care (PRISM) QUERI
Richard L. Roudebush Veterans Affairs Medical Center
Indiana University Center for Health Services & Outcomes Research
Regenstrief Institute, Inc.
Indianapolis, Indiana
on behalf of study co-authors re:
Rattray NA, Flanagan ME, Militello LG, Barach P, Franks Z, Ebright P, Rehman SU,
Gordon HS, Frankel RM
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: End-of-shift handoffs pose a substantial patient safety risk. The transition of care from one doctor to another has been associated with delays in diagnosis and treatment, duplication of tests or treatment and patient discomfort, inappropriate care, medication errors and longer hospital stays with more laboratory testing. Handoff education varies widely in medical schools and residency training programs. Although there have been efforts to improve transfers of care, they have not shown meaningful improvement.
Led for the last decade by Richard Frankel, Ph.D., a senior health scientist at Regenstrief Institute and Indiana University and professor at Indiana University School of Medicine, our team has studied how health practitioners communicate during end-of-shift handoffs. In this current study, funded by VA Health Services and Research Development, we conducted interviews with 35 internal medicine and surgery residents at three VA medical centers about a recent handoff and analyzed the responses. Our team also video-recorded and analyzed more than 150 handoffs.
Published in the Journal of General Internal Medicine, this study explains how the person receiving the handoff can affect the interaction. Medical residents said they changed their delivery based on the doctor or resident who was taking over (i.e., training level, preference for fewer details, day or night shift). We found that handoff communication involves a complex combination of socio-technical information where residents balance relational factors against content and risk. It is not a mechanistic process of merely transferring clinical data but rather is based on learned habits of communication that are context-sensitive and variable, what we refer to as “recipient design”.
In another paper led by Laura Militello, we focus on how residents cognitively prepare for handoffs. In the paper published in The Joint Commission Journal of Quality and Patient Safety®, researchers detailed the tasks involved in cognitively preparing for handoffs. A third paper, published in BMC Medical Education, reports on the limited training that physicians receive during their residency. Residents said they were only partially prepared for enacting handoffs as interns, and clinical experience and enacting handoffs actually taught them the most.
Dr. Rattray[/caption]
Nicholas A. Rattray, Ph.D.
Research Scientist/Investigator
VA HSR&D Center for Health Information and Communication
Implementation Core, Precision Monitoring to Transform Care (PRISM) QUERI
Richard L. Roudebush Veterans Affairs Medical Center
Indiana University Center for Health Services & Outcomes Research
Regenstrief Institute, Inc.
Indianapolis, Indiana
on behalf of study co-authors re:
Rattray NA, Flanagan ME, Militello LG, Barach P, Franks Z, Ebright P, Rehman SU,
Gordon HS, Frankel RM
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: End-of-shift handoffs pose a substantial patient safety risk. The transition of care from one doctor to another has been associated with delays in diagnosis and treatment, duplication of tests or treatment and patient discomfort, inappropriate care, medication errors and longer hospital stays with more laboratory testing. Handoff education varies widely in medical schools and residency training programs. Although there have been efforts to improve transfers of care, they have not shown meaningful improvement.
Led for the last decade by Richard Frankel, Ph.D., a senior health scientist at Regenstrief Institute and Indiana University and professor at Indiana University School of Medicine, our team has studied how health practitioners communicate during end-of-shift handoffs. In this current study, funded by VA Health Services and Research Development, we conducted interviews with 35 internal medicine and surgery residents at three VA medical centers about a recent handoff and analyzed the responses. Our team also video-recorded and analyzed more than 150 handoffs.
Published in the Journal of General Internal Medicine, this study explains how the person receiving the handoff can affect the interaction. Medical residents said they changed their delivery based on the doctor or resident who was taking over (i.e., training level, preference for fewer details, day or night shift). We found that handoff communication involves a complex combination of socio-technical information where residents balance relational factors against content and risk. It is not a mechanistic process of merely transferring clinical data but rather is based on learned habits of communication that are context-sensitive and variable, what we refer to as “recipient design”.
In another paper led by Laura Militello, we focus on how residents cognitively prepare for handoffs. In the paper published in The Joint Commission Journal of Quality and Patient Safety®, researchers detailed the tasks involved in cognitively preparing for handoffs. A third paper, published in BMC Medical Education, reports on the limited training that physicians receive during their residency. Residents said they were only partially prepared for enacting handoffs as interns, and clinical experience and enacting handoffs actually taught them the most.




 Dr. Zhu[/caption]
Wenjia Zhu, PhD.
Marshall J. Seidman Fellow
Department of Health Care Policy
Harvard Medical School
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The current opioid epidemic continues to cause deaths and tremendous suffering in the United States, driven in large part by overuse of prescription opioids. Of special concern are new opioid prescriptions, i.e. opioids given to patients who have not used opioids before, which research tells us are an important gateway to long-term opioid use, misuse, overdoes and death. Recently, in their efforts to curb over prescribing of opioids, the CDC issued guidelines (December 2015 in draft form; March 2016 in final version) to encourage opioid prescribers to limit the use, duration and dose of opioids, particularly opioids to first-time users. Despite these, little is known about the prescribing of opioids to first-time users on a national scale, particularly among commercially insured patients.
In this study, we examined national monthly trends in the rate at which opioid therapy was started among commercially insured patients. Using administrative claims from Blue Cross Blue Shield Association commercial insurers from 2012 to 2017, we analyzed more than 86 million commercially insured patients across the United States.
Dr. Zhu[/caption]
Wenjia Zhu, PhD.
Marshall J. Seidman Fellow
Department of Health Care Policy
Harvard Medical School
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The current opioid epidemic continues to cause deaths and tremendous suffering in the United States, driven in large part by overuse of prescription opioids. Of special concern are new opioid prescriptions, i.e. opioids given to patients who have not used opioids before, which research tells us are an important gateway to long-term opioid use, misuse, overdoes and death. Recently, in their efforts to curb over prescribing of opioids, the CDC issued guidelines (December 2015 in draft form; March 2016 in final version) to encourage opioid prescribers to limit the use, duration and dose of opioids, particularly opioids to first-time users. Despite these, little is known about the prescribing of opioids to first-time users on a national scale, particularly among commercially insured patients.
In this study, we examined national monthly trends in the rate at which opioid therapy was started among commercially insured patients. Using administrative claims from Blue Cross Blue Shield Association commercial insurers from 2012 to 2017, we analyzed more than 86 million commercially insured patients across the United States.



 Dr. Leung[/caption]
Alexander A. Leung, MD, MPH
Department of Community Health Sciences
Department of Medicine
University of Calgary
Calgary, Alberta, Canada
MedicalResearch.com: What is the background for this study?
Response: The 2017 American College of Cardiology and American Heart Association (ACC/AHA) blood pressure guidelines redefined hypertension according to a blood pressure cutoff of ≥130/80 mm Hg, compared to the traditional cutoff of ≥140/90 mm Hg.
Dr. Leung[/caption]
Alexander A. Leung, MD, MPH
Department of Community Health Sciences
Department of Medicine
University of Calgary
Calgary, Alberta, Canada
MedicalResearch.com: What is the background for this study?
Response: The 2017 American College of Cardiology and American Heart Association (ACC/AHA) blood pressure guidelines redefined hypertension according to a blood pressure cutoff of ≥130/80 mm Hg, compared to the traditional cutoff of ≥140/90 mm Hg.





 Dr. Gyamfi-Bannerman[/caption]
Cynthia Gyamfi-Bannerman, MD, MSc
Ellen Jacobson Levine and Eugene Jacobson
Professor of Women's Health in Obstetrics and Gynecology
Director, Maternal-Fetal Medicine Fellowship Program
Co-Director, CUMC Preterm Birth Prevention Center
Columbia University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2016 our group published the findings of the Antenatal Late Preterm Steroids (ALPS) trial in the NEJM. We found that administration of antenatal corticosteroids to women at high risk for delivery from 34-36 weeks decreased breathing problems in their neonates. This treatment had been traditionally only given at less than 34 weeks.
The current paper is a cost analysis of that trial. We found that the treatment was also cost effective. From a cost perspective treatment was both low cost and highly effective (the options are low cost, low effect/low cost/high effect, high cost/low effect, high cost/high effect).
Dr. Gyamfi-Bannerman[/caption]
Cynthia Gyamfi-Bannerman, MD, MSc
Ellen Jacobson Levine and Eugene Jacobson
Professor of Women's Health in Obstetrics and Gynecology
Director, Maternal-Fetal Medicine Fellowship Program
Co-Director, CUMC Preterm Birth Prevention Center
Columbia University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2016 our group published the findings of the Antenatal Late Preterm Steroids (ALPS) trial in the NEJM. We found that administration of antenatal corticosteroids to women at high risk for delivery from 34-36 weeks decreased breathing problems in their neonates. This treatment had been traditionally only given at less than 34 weeks.
The current paper is a cost analysis of that trial. We found that the treatment was also cost effective. From a cost perspective treatment was both low cost and highly effective (the options are low cost, low effect/low cost/high effect, high cost/low effect, high cost/high effect). 

 Dr. Rhee[/caption]
Chanu Rhee, MD,MPH
Assistant Professor of Population Medicine
Harvard Medical School / Harvard Pilgrim Health Care Institute
Assistant Hospital Epidemiologist
Brigham and Women’s Hospital
MedicalResearch.com: What is the background for this study?
Response: Sepsis is the body’s reaction to a serious infection that results a cascade of inflammation in the body and organ dysfunction, such as low blood pressure, confusion, or failure of the lungs, kidneys, or liver. Sepsis is a major cause of death, disability, and cost in the U.S. and around the world. Growing recognition of this problem has led to numerous sepsis performance improvement initiatives in hospitals around the country. Some of these efforts have also been catalyzed by high-profile tragic cases of missed sepsis leading to death, which may have contributed to a perception that most sepsis deaths are preventable if doctors and hospitals were only better at recognizing it.
However, the extent to which sepsis-related deaths might be preventable with better hospital-based care is unknown. In my own experience as a critical care physician, a lot of sepsis patients we treat are extremely sick and even when they receive timely and optimal medical care, many do not survive. This led myself and my colleagues to conduct this study to better understand what types of patients are dying from sepsis and how preventable these deaths might be.
Dr. Rhee[/caption]
Chanu Rhee, MD,MPH
Assistant Professor of Population Medicine
Harvard Medical School / Harvard Pilgrim Health Care Institute
Assistant Hospital Epidemiologist
Brigham and Women’s Hospital
MedicalResearch.com: What is the background for this study?
Response: Sepsis is the body’s reaction to a serious infection that results a cascade of inflammation in the body and organ dysfunction, such as low blood pressure, confusion, or failure of the lungs, kidneys, or liver. Sepsis is a major cause of death, disability, and cost in the U.S. and around the world. Growing recognition of this problem has led to numerous sepsis performance improvement initiatives in hospitals around the country. Some of these efforts have also been catalyzed by high-profile tragic cases of missed sepsis leading to death, which may have contributed to a perception that most sepsis deaths are preventable if doctors and hospitals were only better at recognizing it.
However, the extent to which sepsis-related deaths might be preventable with better hospital-based care is unknown. In my own experience as a critical care physician, a lot of sepsis patients we treat are extremely sick and even when they receive timely and optimal medical care, many do not survive. This led myself and my colleagues to conduct this study to better understand what types of patients are dying from sepsis and how preventable these deaths might be. 
 Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
 Dr. McCrory[/caption]
Megan A McCrory, PhD, FTOS
Research Associate Professor
Dept of Health Sciences
Sargent College of Health and Rehabilitation Sciences
Boston University 02215
MedicalResearch.com: What is the background for this study?
Response: The prevalence of overweight and obesity has increased in the US, along with documented increases in portion size in the food supply. Fast food is popular, making up about 11% of adult daily calorie intake in the US, and over 1/3 of U.S. adults eat at fast food establishments on any given day. We therefore sought to examine changes in portion size, calories, and selected nutrients in fast-food entree, side, and dessert menu items across the years 1986, 1991, and 2016.
Dr. McCrory[/caption]
Megan A McCrory, PhD, FTOS
Research Associate Professor
Dept of Health Sciences
Sargent College of Health and Rehabilitation Sciences
Boston University 02215
MedicalResearch.com: What is the background for this study?
Response: The prevalence of overweight and obesity has increased in the US, along with documented increases in portion size in the food supply. Fast food is popular, making up about 11% of adult daily calorie intake in the US, and over 1/3 of U.S. adults eat at fast food establishments on any given day. We therefore sought to examine changes in portion size, calories, and selected nutrients in fast-food entree, side, and dessert menu items across the years 1986, 1991, and 2016.





 Dr. Mikkola[/caption]
Tomi Mikkola MD
Associate Professor
Helsinki University Hospital
Department of Obstetrics and Gynecology
Helsinki, Finland
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In Finland we have perhaps the most comprehensive and reliable medical registers in the world. Thus, with my research group I have conducted various large studies evaluating association of postmenopausal hormone therapy use and various major diseases (see e.g. the references in the B;MJ paper). There has been various smaller studies indicating that hormone therapy might be protective for all kinds of dementias, also Alzheimer’s disease.
However, we have quite recently shown that hormone therapy seems to lower the mortality risk of vascular dementia but not Alzheimer’s disease (Mikkola TS et al. J Clin Endocrinol Metab 2017;102:870-7). Now in this upcoming BMJ-paper we report in a very large case-control study (83 688 women with Alzheimer’s disease and same number of control women without the disease) that systemic hormone therapy was associated with a 9-17% increased risk of Alzheimer’s disease.
Furthermore, this risk increase is particularly in women using hormone therapy long, for more than 10 years. This was somewhat surprising finding, but it underlines the fact that mechanisms behind Alzheimer’s disease are likely quite different than in vascular dementia, where the risk factors are similar as in cardiovascular disease. We have also shown how hormone therapy protects against cardiovascular disease, particularly in women who initiate hormone therapy soon after menopause.
Dr. Mikkola[/caption]
Tomi Mikkola MD
Associate Professor
Helsinki University Hospital
Department of Obstetrics and Gynecology
Helsinki, Finland
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In Finland we have perhaps the most comprehensive and reliable medical registers in the world. Thus, with my research group I have conducted various large studies evaluating association of postmenopausal hormone therapy use and various major diseases (see e.g. the references in the B;MJ paper). There has been various smaller studies indicating that hormone therapy might be protective for all kinds of dementias, also Alzheimer’s disease.
However, we have quite recently shown that hormone therapy seems to lower the mortality risk of vascular dementia but not Alzheimer’s disease (Mikkola TS et al. J Clin Endocrinol Metab 2017;102:870-7). Now in this upcoming BMJ-paper we report in a very large case-control study (83 688 women with Alzheimer’s disease and same number of control women without the disease) that systemic hormone therapy was associated with a 9-17% increased risk of Alzheimer’s disease.
Furthermore, this risk increase is particularly in women using hormone therapy long, for more than 10 years. This was somewhat surprising finding, but it underlines the fact that mechanisms behind Alzheimer’s disease are likely quite different than in vascular dementia, where the risk factors are similar as in cardiovascular disease. We have also shown how hormone therapy protects against cardiovascular disease, particularly in women who initiate hormone therapy soon after menopause.




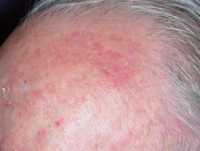
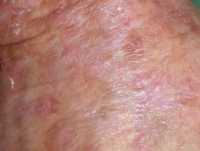
 Dr. Del Rosso[/caption]
Dr. James Q. Del Rosso, D.O., FAOCD, Dermatologist
Research Director and Principal Investigator
Del Rosso Dermatology Research Center, Las Vegas, NV
Galderma Consultant
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Del Rosso[/caption]
Dr. James Q. Del Rosso, D.O., FAOCD, Dermatologist
Research Director and Principal Investigator
Del Rosso Dermatology Research Center, Las Vegas, NV
Galderma Consultant
MedicalResearch.com: What is the background for this study? What are the main findings?