MedicalResearch.com Interview with:
[caption id="attachment_53477" align="alignleft" width="200"]

Dr. Wallach[/caption]
Joshua D. Wallach, MS, PhD
Assistant Professor of Epidemiology (Environmental Health Sciences)
Yale School of Public Health
New Haven, CT
MedicalResearch.com: What is the background for this study?
Response: Over the past few years, there has been growing interest in the potential health benefits of cannabidiol (CBD), a chemical compound in cannabis. Although only one CBD-derived prescription drug has been approved by the US Food and Drug Administration (FDA) for the treatment of epilepsy, I recently started seeing products containing CBD advertised and sold across the US (e.g. CBD in foods, beverages, dietary supplements, and cosmetics). I noticed that many of these products were being marketed with unproven claims to prevent, cure, and treat various conditions, and became interested in learning more about the research supporting the use of CBD, the potential for misleading claims, and impact that the CBD-industry may be having on research that is being generated and disseminated to the public.
Research funding sources and other author conflicts of interests (e.g. consulting fees, honoraria, travel expenses) can influence the way that research is designed, conducted, and reported. Previous studies have consistently demonstrated associations between authors' conflicts of interest and proindustry conclusions in clinical research.
Given the growing number of companies invested in CBD's commercial success, we decided to analyze the disclosed funding sources, conflicts of interest statements, author employment details, and CBD-related conclusions in a large sample of published articles on the characteristics, use, and therapeutic effects of cannabidiol.







 Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
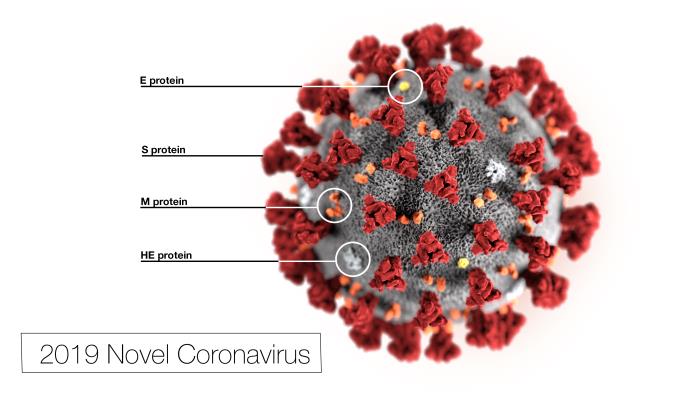
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.
Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.



 Dr. Jeffrey Smith[/caption]
Jeffrey R. Smith, MD PhD
Department of Medicine, Division of Genetic Medicine
Vanderbilt-Ingram Cancer Center, and Vanderbilt Genetics Institute
Vanderbilt University Medical Center
Medical Research Service
Tennessee Valley Healthcare System, Veterans Administration
Nashville, TN
MedicalResearch.com: What is the background for this study?
Response: Roughly 20% of men with prostate cancer have a family history of the disease, and 5% meet criteria for hereditary prostate cancer. Although prostate cancer has the greatest heritability of all common cancers (twice that of breast cancer), extensive heterogeneity of its inherited causes has presented a considerable obstacle for traditional pedigree-based genetic investigative approaches. Inherited causes across, as well as within families are diverse.
This study introduced a new familial case-control study design that uses extent of family history as a proxy for genetic burden. It compared a large number of men with prostate cancer, each from a separate family with a strong history of the disease, to screened men with no personal or family history. The study comprehensively deconstructs how the 8q24 chromosomal region impacts risk of hereditary prostate cancer, introducing several new analytical approaches. The locus had been known to alter risk of prostate, breast, colon, ovarian, and numerous additional cancers.
Dr. Jeffrey Smith[/caption]
Jeffrey R. Smith, MD PhD
Department of Medicine, Division of Genetic Medicine
Vanderbilt-Ingram Cancer Center, and Vanderbilt Genetics Institute
Vanderbilt University Medical Center
Medical Research Service
Tennessee Valley Healthcare System, Veterans Administration
Nashville, TN
MedicalResearch.com: What is the background for this study?
Response: Roughly 20% of men with prostate cancer have a family history of the disease, and 5% meet criteria for hereditary prostate cancer. Although prostate cancer has the greatest heritability of all common cancers (twice that of breast cancer), extensive heterogeneity of its inherited causes has presented a considerable obstacle for traditional pedigree-based genetic investigative approaches. Inherited causes across, as well as within families are diverse.
This study introduced a new familial case-control study design that uses extent of family history as a proxy for genetic burden. It compared a large number of men with prostate cancer, each from a separate family with a strong history of the disease, to screened men with no personal or family history. The study comprehensively deconstructs how the 8q24 chromosomal region impacts risk of hereditary prostate cancer, introducing several new analytical approaches. The locus had been known to alter risk of prostate, breast, colon, ovarian, and numerous additional cancers.