Author Interviews, Critical Care - Intensive Care - ICUs, Infections, PLoS / 17.09.2020
Sepsis: Monoclonal Antibody Procizumab Improves Cardiac Function in Animal Model Study
MedicalResearch.com Interview with:
[caption id="attachment_55417" align="alignleft" width="150"] Prof. Mebazaa[/caption]
Prof. Alexandre Mebazaa
Head of the Department of Anesthesia and Critical Care
Hôpital Lariboisiere and of the Research group MASCOT supported by Inserm and the Université de Paris (Paris, France).
Prof. Mebazaa is the principle investigator of the recently published preclinical experiments on Procizumab, a potent, pre-clinical drug candidate targeting DPP3 in patients with acute mycardial depression.
MedicalResearch.com: What is the background for this study? What is the significance of DPP3?
Response: The global burden of sepsis counts for one in three deaths world-wide. Recent findings have shown that circulating Dipeptidyl Pepidase 3 (cDPP3) is elevated in critical patients, including cardiogenic shock and septic patients, with the highest DPP3 blood levels found in non-survivors.
Dipeptidyl Peptidase 3 (DPP3) is an intracellular peptidase that is released into the bloodstream upon cell injury and death, where it inactivates many circulating peptides including angiotensin II. This process likely leads to cardiac depression. Procizumab is a humanized monoclonal antibody in preclinical development that targets and modulates DPP3. The aim of the current study was to evaluate the benefits of inhibiting circulating DPP3 by Procizumab in a preclinical model of sepsis-induced myocardial depression.
Prof. Mebazaa[/caption]
Prof. Alexandre Mebazaa
Head of the Department of Anesthesia and Critical Care
Hôpital Lariboisiere and of the Research group MASCOT supported by Inserm and the Université de Paris (Paris, France).
Prof. Mebazaa is the principle investigator of the recently published preclinical experiments on Procizumab, a potent, pre-clinical drug candidate targeting DPP3 in patients with acute mycardial depression.
MedicalResearch.com: What is the background for this study? What is the significance of DPP3?
Response: The global burden of sepsis counts for one in three deaths world-wide. Recent findings have shown that circulating Dipeptidyl Pepidase 3 (cDPP3) is elevated in critical patients, including cardiogenic shock and septic patients, with the highest DPP3 blood levels found in non-survivors.
Dipeptidyl Peptidase 3 (DPP3) is an intracellular peptidase that is released into the bloodstream upon cell injury and death, where it inactivates many circulating peptides including angiotensin II. This process likely leads to cardiac depression. Procizumab is a humanized monoclonal antibody in preclinical development that targets and modulates DPP3. The aim of the current study was to evaluate the benefits of inhibiting circulating DPP3 by Procizumab in a preclinical model of sepsis-induced myocardial depression.
 Prof. Mebazaa[/caption]
Prof. Alexandre Mebazaa
Head of the Department of Anesthesia and Critical Care
Hôpital Lariboisiere and of the Research group MASCOT supported by Inserm and the Université de Paris (Paris, France).
Prof. Mebazaa is the principle investigator of the recently published preclinical experiments on Procizumab, a potent, pre-clinical drug candidate targeting DPP3 in patients with acute mycardial depression.
MedicalResearch.com: What is the background for this study? What is the significance of DPP3?
Response: The global burden of sepsis counts for one in three deaths world-wide. Recent findings have shown that circulating Dipeptidyl Pepidase 3 (cDPP3) is elevated in critical patients, including cardiogenic shock and septic patients, with the highest DPP3 blood levels found in non-survivors.
Dipeptidyl Peptidase 3 (DPP3) is an intracellular peptidase that is released into the bloodstream upon cell injury and death, where it inactivates many circulating peptides including angiotensin II. This process likely leads to cardiac depression. Procizumab is a humanized monoclonal antibody in preclinical development that targets and modulates DPP3. The aim of the current study was to evaluate the benefits of inhibiting circulating DPP3 by Procizumab in a preclinical model of sepsis-induced myocardial depression.
Prof. Mebazaa[/caption]
Prof. Alexandre Mebazaa
Head of the Department of Anesthesia and Critical Care
Hôpital Lariboisiere and of the Research group MASCOT supported by Inserm and the Université de Paris (Paris, France).
Prof. Mebazaa is the principle investigator of the recently published preclinical experiments on Procizumab, a potent, pre-clinical drug candidate targeting DPP3 in patients with acute mycardial depression.
MedicalResearch.com: What is the background for this study? What is the significance of DPP3?
Response: The global burden of sepsis counts for one in three deaths world-wide. Recent findings have shown that circulating Dipeptidyl Pepidase 3 (cDPP3) is elevated in critical patients, including cardiogenic shock and septic patients, with the highest DPP3 blood levels found in non-survivors.
Dipeptidyl Peptidase 3 (DPP3) is an intracellular peptidase that is released into the bloodstream upon cell injury and death, where it inactivates many circulating peptides including angiotensin II. This process likely leads to cardiac depression. Procizumab is a humanized monoclonal antibody in preclinical development that targets and modulates DPP3. The aim of the current study was to evaluate the benefits of inhibiting circulating DPP3 by Procizumab in a preclinical model of sepsis-induced myocardial depression.





 Dr. Obisesan[/caption]
Funmi (Abiru) Obisesan, MD, MPH
Postdoctoral Fellow
Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease
Postdoctoral Fellow
Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease
MedicalResearch.com: What is the background for this study?
Response: E-cigarettes are conceived by many individuals to be less harmful than combustible cigarettes, resulting in their use among young individuals and other vulnerable subpopulations. The recent outbreak of EVALI (e-cigarette, or vaping, product-use associated lung injuries) which was directly associated with e-cigarette use, and the rapidly evolving e-cigarette market, as well as regulations concerning them necessitates the need for up-to-date analyses of e-cigarette use trends.
Dr. Obisesan[/caption]
Funmi (Abiru) Obisesan, MD, MPH
Postdoctoral Fellow
Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease
Postdoctoral Fellow
Johns Hopkins Ciccarone Center for the Prevention of Cardiovascular Disease
MedicalResearch.com: What is the background for this study?
Response: E-cigarettes are conceived by many individuals to be less harmful than combustible cigarettes, resulting in their use among young individuals and other vulnerable subpopulations. The recent outbreak of EVALI (e-cigarette, or vaping, product-use associated lung injuries) which was directly associated with e-cigarette use, and the rapidly evolving e-cigarette market, as well as regulations concerning them necessitates the need for up-to-date analyses of e-cigarette use trends. 






 Dr. Spitzer[/caption]
Sarabeth Spitzer, MD
Co-Chair of Board, Scrubs Addressing the Firearm Epidemic (SAFE)
Department of Surgery, Brigham and Women’s Hospital
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Firearm injury is a significant cause of morbidity and mortality in the United States, resulting in almost 40,000 deaths annually in the United States, but very little is known about the epidemiology of nonfatal firearm injuries. Nonfatal firearm injuries can have significant long-term morbidity and are associated with significant cost. We found that there were over 81,000 nonfatal firearm injuries in California over the study period. Over the period, there was a decrease in nonfatal firearm injuries by 38.1%, driven primarily by a decrease in assault injuries.
Dr. Spitzer[/caption]
Sarabeth Spitzer, MD
Co-Chair of Board, Scrubs Addressing the Firearm Epidemic (SAFE)
Department of Surgery, Brigham and Women’s Hospital
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Firearm injury is a significant cause of morbidity and mortality in the United States, resulting in almost 40,000 deaths annually in the United States, but very little is known about the epidemiology of nonfatal firearm injuries. Nonfatal firearm injuries can have significant long-term morbidity and are associated with significant cost. We found that there were over 81,000 nonfatal firearm injuries in California over the study period. Over the period, there was a decrease in nonfatal firearm injuries by 38.1%, driven primarily by a decrease in assault injuries. 
 Dr. Shoaff[/caption]
Jessica Shoaff, MPH, PhD
Postdoctoral Research Fellow and
[caption id="attachment_55259" align="alignleft" width="150"]
Dr. Shoaff[/caption]
Jessica Shoaff, MPH, PhD
Postdoctoral Research Fellow and
[caption id="attachment_55259" align="alignleft" width="150"] Dr. Korrick[/caption]
Susan A. Korrick, MD
Pulmonary and Critical Care
Assistant Professor in the Department of Environmental Health
Harvard Medical School · Harvard T. H. Chan School of Public Health
Brigham and Women's Hospital
Channing Laboratory Boston, MA 02115
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Our study posed the question: Do teenagers’ exposures to chemicals that are often found in consumer products increase behaviors that are common among individuals diagnosed with Attention-Deficit/Hyperactivity Disorder (ADHD)? Our results suggest that teenagers exposed to chemicals often found in consumer products (particularly phthalates) may have increased behaviors that are common among individuals diagnosed with ADHD. However, we did not study the diagnosis of ADHD (most of our study teens did not have ADHD). This means our results cannot answer the question of whether these chemical exposures increase the likelihood of being diagnosed with ADHD. Also, in our study design, chemical exposures and ADHD-related behaviors were measured at the same time, so it is not possible to know with certainty whether the chemical exposures altered behavior or behavior altered chemical exposures.
Dr. Korrick[/caption]
Susan A. Korrick, MD
Pulmonary and Critical Care
Assistant Professor in the Department of Environmental Health
Harvard Medical School · Harvard T. H. Chan School of Public Health
Brigham and Women's Hospital
Channing Laboratory Boston, MA 02115
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Our study posed the question: Do teenagers’ exposures to chemicals that are often found in consumer products increase behaviors that are common among individuals diagnosed with Attention-Deficit/Hyperactivity Disorder (ADHD)? Our results suggest that teenagers exposed to chemicals often found in consumer products (particularly phthalates) may have increased behaviors that are common among individuals diagnosed with ADHD. However, we did not study the diagnosis of ADHD (most of our study teens did not have ADHD). This means our results cannot answer the question of whether these chemical exposures increase the likelihood of being diagnosed with ADHD. Also, in our study design, chemical exposures and ADHD-related behaviors were measured at the same time, so it is not possible to know with certainty whether the chemical exposures altered behavior or behavior altered chemical exposures. 

 Dr. Wick[/caption]
Cameron C. Wick, MD
Assistant Professor, Otology/Neurotology
Washington University School of Medicine
St. Louis, MO
MedicalResearch.com: What do you see as the primary message of your findings for the general public?
Response: Older adults not satisfied with their hearing aids achieved clinically meaningful improvement in both hearing and quality of life with a cochlear implant compared to an optimized bilateral hearing aid condition.
MedicalResearch.com: Do you see your findings as changing the way older adults with hearing loss are managed?
Response: Yes and partially because this study is unique in its design and the outcomes that were measured. Specifically the study is a prospective, multicenter clinical trial conducted at 13 locations across the United States. All patients were setup with a 30-day optimized hearing aid experience before cochlear implantation (context: sometimes hearing aids are not appropriately optimized so baseline testing may not reflect the "best" that hearing aids can do). This study assesses both hearing data as well as quality of life data before and 6-months after cochlear implantation. After implantation patients were tested in both the unilateral (cochlear implant alone) and bimodal (cochlear implant plus hearing aid in the opposite ear) conditions. My paper is a subanalysis of adults 65 years and older (range 65 - 91 years) enrolled in the clinical trial. The principal investigator of the clinical trial is Dr. Craig Buchman. Dr. Buchman and myself are at Washington University in St. Louis which was the lead center for the clinical trial.
The findings of the study are meaningful because they demonstrate clear superiority of cochlear implants over hearing aids in many key areas, such as understanding speech, hearing in background noise, and ability to communicate. Hearing loss, which becomes more prevalent as we age, can negatively impact communication leading to social isolation, depression, frustration, and possibly cognitive decline. This study highlights that if patients are not satisfied with their hearing aid performance then they should be referred to a center that can evaluate for cochlear implantation. Cochlear implant indications have evolved considerably since they were first FDA approved in 1984. This study emphasizes that patients do not have to be profoundly deaf to experience significant hearing and social benefits from cochlear implants. Also, it demonstrates that cochlear implant surgery is well tolerated even as adults age and acquire other health ailments.
Dr. Wick[/caption]
Cameron C. Wick, MD
Assistant Professor, Otology/Neurotology
Washington University School of Medicine
St. Louis, MO
MedicalResearch.com: What do you see as the primary message of your findings for the general public?
Response: Older adults not satisfied with their hearing aids achieved clinically meaningful improvement in both hearing and quality of life with a cochlear implant compared to an optimized bilateral hearing aid condition.
MedicalResearch.com: Do you see your findings as changing the way older adults with hearing loss are managed?
Response: Yes and partially because this study is unique in its design and the outcomes that were measured. Specifically the study is a prospective, multicenter clinical trial conducted at 13 locations across the United States. All patients were setup with a 30-day optimized hearing aid experience before cochlear implantation (context: sometimes hearing aids are not appropriately optimized so baseline testing may not reflect the "best" that hearing aids can do). This study assesses both hearing data as well as quality of life data before and 6-months after cochlear implantation. After implantation patients were tested in both the unilateral (cochlear implant alone) and bimodal (cochlear implant plus hearing aid in the opposite ear) conditions. My paper is a subanalysis of adults 65 years and older (range 65 - 91 years) enrolled in the clinical trial. The principal investigator of the clinical trial is Dr. Craig Buchman. Dr. Buchman and myself are at Washington University in St. Louis which was the lead center for the clinical trial.
The findings of the study are meaningful because they demonstrate clear superiority of cochlear implants over hearing aids in many key areas, such as understanding speech, hearing in background noise, and ability to communicate. Hearing loss, which becomes more prevalent as we age, can negatively impact communication leading to social isolation, depression, frustration, and possibly cognitive decline. This study highlights that if patients are not satisfied with their hearing aid performance then they should be referred to a center that can evaluate for cochlear implantation. Cochlear implant indications have evolved considerably since they were first FDA approved in 1984. This study emphasizes that patients do not have to be profoundly deaf to experience significant hearing and social benefits from cochlear implants. Also, it demonstrates that cochlear implant surgery is well tolerated even as adults age and acquire other health ailments. 


 Dr. Jimenez[/caption]
Monik Carmen Jimenez, Sc.D
Assistant Professor of Medicine
Brigham and Women's Hospital
MedicalResearch.com: What is the background for this study?
Response: We wanted to get a comprehensive picture of the epidemiology of COVID-19 in carceral facilities that included jails and was not restricted solely to prisons. We utilized publicly available data collected in Massachusetts, pursuant to a court order. These data included prison and jail systems and were used to calculate rates of confirmed cases of COVID-19 and testing rates among incarcerated individuals. We were also able to compare those to changes in the population size within each system.
Dr. Jimenez[/caption]
Monik Carmen Jimenez, Sc.D
Assistant Professor of Medicine
Brigham and Women's Hospital
MedicalResearch.com: What is the background for this study?
Response: We wanted to get a comprehensive picture of the epidemiology of COVID-19 in carceral facilities that included jails and was not restricted solely to prisons. We utilized publicly available data collected in Massachusetts, pursuant to a court order. These data included prison and jail systems and were used to calculate rates of confirmed cases of COVID-19 and testing rates among incarcerated individuals. We were also able to compare those to changes in the population size within each system.




 Dr. Reyes Gil[/caption]
Dr. Reyes Gil[/caption]


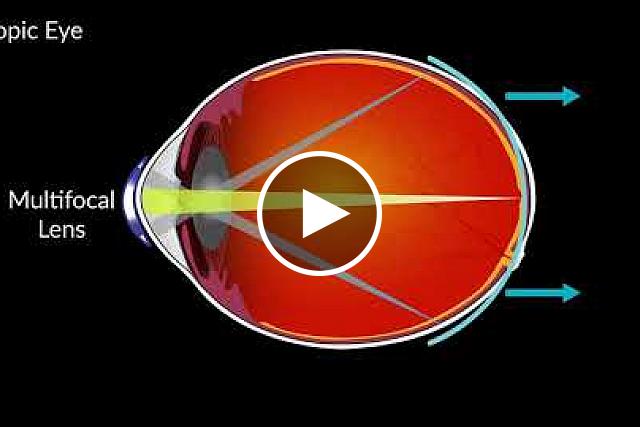
 Dr. Walline[/caption]
Jeffrey J. Walline, OD PhD
Associate Dean for Research
The Ohio State University
Columbus, OH 43210-1240
MedicalResearch.com: What is the background for this study?
Response: Greater amounts of nearsightedness are related to higher risks of sight-threatening complications in adulthood, so anything we can do to slow the progression of nearsightedness in childhood can have meaningful benefits in the future.
As the prevalence of nearsightedness increases worldwide and affects approximately 1/3 of the people in the United States, a treatment that provides clear vision AND slows the progression of nearsightedness can have a profound effect.
Dr. Walline[/caption]
Jeffrey J. Walline, OD PhD
Associate Dean for Research
The Ohio State University
Columbus, OH 43210-1240
MedicalResearch.com: What is the background for this study?
Response: Greater amounts of nearsightedness are related to higher risks of sight-threatening complications in adulthood, so anything we can do to slow the progression of nearsightedness in childhood can have meaningful benefits in the future.
As the prevalence of nearsightedness increases worldwide and affects approximately 1/3 of the people in the United States, a treatment that provides clear vision AND slows the progression of nearsightedness can have a profound effect.