Author Interviews, Hematology / 19.12.2019
Rozanolixizumab, an Anti-FcRn Antibody for ITP: Primary Immune Thrombocytopenia
MedicalResearch.com Interview with:
[caption id="attachment_52512" align="alignleft" width="80"] Dr. Robak[/caption]
Tadeusz Robak MD, Ph.D.
Professor of Hematology
Medical University of Lodz, Poland
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Robak[/caption]
Tadeusz Robak MD, Ph.D.
Professor of Hematology
Medical University of Lodz, Poland
MedicalResearch.com: What is the background for this study? What are the main findings?
 Dr. Robak[/caption]
Tadeusz Robak MD, Ph.D.
Professor of Hematology
Medical University of Lodz, Poland
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Robak[/caption]
Tadeusz Robak MD, Ph.D.
Professor of Hematology
Medical University of Lodz, Poland
MedicalResearch.com: What is the background for this study? What are the main findings?
- Rozanolixizumab is an advanced SC anti-neonatal FcRn therapy currently in clinical development which has the potential to provide a targeted, convenient option to optimize individualized patient care.
- The rozanolixizumab Phase II (TP0001) study was specifically designed to explore multiple dose regimens in order to inform the dosing strategy for further development in primary immune thrombocytopenia (ITP). Patients received either a single dose (1 x 15 mg/kg or 1 x 20 mg/kg) or multiple doses (5 x 4 mg/kg, 3 x 7 mg/kg, 2 x 10 mg/kg) of subcutaneous (SC) rozanolixizumab. The total dose was similar in all treatment groups, ranging from 15 to 21 mg/kg.
- Rozanolixizumab was well tolerated by patients with primary ITP across all dose groups, consistent with previous rozanolixizumab studies. Additionally, improved platelet counts and reduced immunoglobin G (IgG) levels were seen at all doses and regimens of rozanolixizumab treatment, with higher response rate, higher percentage of responders and shorter time to response achieved by the 1 x 15 mg/kg, 1 x 20 mg/kg and the 3 x 7 mg/kg rozanolixizumab dose groups. These safety, tolerability and efficacy data support the Phase III development in patients with primary ITP.





 Dr. Aaron Elliott[/caption]
Dr. Aaron Elliott, PhD
CEO
Dr. Aaron Elliott[/caption]
Dr. Aaron Elliott, PhD
CEO




 Dr. Chen[/caption]
Tiffany Won-Shau Chen MD
Internal Medicine Residency
Mount Sinai Beth Israel
MedicalResearch.com: What is the background for this study?
Response: The research I presented on details a randomized, prospective study done to evaluate whether it would be feasible and effective to implement a yoga program for breast cancer patients receiving chemotherapy that could reduce patients' chemotherapy-related symptoms and improve their quality of life.
50 patients were recruited, half of whom underwent a 12-week long yoga program with weekly courses, while the other half did not participate in the program.
Surveys were completed at baseline, 6 weeks, and 12 weeks assessing patients' functional wellbeing, sleep quality, and anxiety/depression levels.
Dr. Chen[/caption]
Tiffany Won-Shau Chen MD
Internal Medicine Residency
Mount Sinai Beth Israel
MedicalResearch.com: What is the background for this study?
Response: The research I presented on details a randomized, prospective study done to evaluate whether it would be feasible and effective to implement a yoga program for breast cancer patients receiving chemotherapy that could reduce patients' chemotherapy-related symptoms and improve their quality of life.
50 patients were recruited, half of whom underwent a 12-week long yoga program with weekly courses, while the other half did not participate in the program.
Surveys were completed at baseline, 6 weeks, and 12 weeks assessing patients' functional wellbeing, sleep quality, and anxiety/depression levels. Dr. Cohen[/caption]
Pieter A. Cohen, MD
Department of Medicine, Cambridge Health Alliance
Somerville, Massachusetts
Harvard Medical School
Boston, Massachusetts
MedicalResearch.com: What is the background for this study?
Response: There has been increasing interest in the use of over-the-counter supplements to help improve memory and cognitive function. However, prior
Dr. Cohen[/caption]
Pieter A. Cohen, MD
Department of Medicine, Cambridge Health Alliance
Somerville, Massachusetts
Harvard Medical School
Boston, Massachusetts
MedicalResearch.com: What is the background for this study?
Response: There has been increasing interest in the use of over-the-counter supplements to help improve memory and cognitive function. However, prior 
 Dr. Ritch[/caption]
Robert Ritch, MD, FACS, FARVO
Shelley and Steven Einhorn Distinguished Chair
Professor of Ophthalmology, Surgeon Director Emeritus
Chief, Glaucoma Services Emeritus
The New York Eye and Ear Infirmary of Mount Sinai
New York, NY 10003
Founder, Medical Director and
Chairman, Scientific Advisory Board
The Glaucoma Foundation
MedicalResearch.com: What is the background for this study?
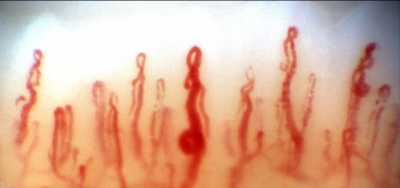
Response: Nailfold capillaroscopy (NFC), long used in rheumatology is a new approach to investigation of glaucoma.
Posterior to the nailbed and just anterior to the proximal nailfold is the cuticle, which has no structural elements visible to the naked eye. NFC is a non-invasive imaging modality that provides a highly magnified view of the capillaries at the nailfold of digits. It has also been used in ophthalmology to show morphological changes at the nailfold capillaries of POAG and XFG/XFS patients, helping to confirm the systemic nature of these diseases.
[caption id="attachment_52469" align="alignleft" width="400"]
Dr. Ritch[/caption]
Robert Ritch, MD, FACS, FARVO
Shelley and Steven Einhorn Distinguished Chair
Professor of Ophthalmology, Surgeon Director Emeritus
Chief, Glaucoma Services Emeritus
The New York Eye and Ear Infirmary of Mount Sinai
New York, NY 10003
Founder, Medical Director and
Chairman, Scientific Advisory Board
The Glaucoma Foundation
MedicalResearch.com: What is the background for this study?
Response: Nailfold capillaroscopy (NFC), long used in rheumatology is a new approach to investigation of glaucoma.
Posterior to the nailbed and just anterior to the proximal nailfold is the cuticle, which has no structural elements visible to the naked eye. NFC is a non-invasive imaging modality that provides a highly magnified view of the capillaries at the nailfold of digits. It has also been used in ophthalmology to show morphological changes at the nailfold capillaries of POAG and XFG/XFS patients, helping to confirm the systemic nature of these diseases.
[caption id="attachment_52469" align="alignleft" width="400"]