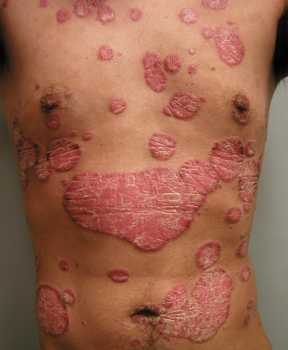
Author Interviews, Heart Disease, Menopause, Osteoporosis / 12.05.2021
Low Bone Mineral Density Linked to Heart Disease in Women
MedicalResearch.com Interview with:
Yeonyee E. Yoon, MD, PhD
Associate Professor
Division of Cardiology, Cardiovascular Center
Seoul National University Bundang Hospital
South Korea
MedicalResearch.com: What is the background for this study?
Response: Although atherosclerotic cardiovascular disease (ASCVD) has been traditionally considered to affect men predominantly, it is nearly common in women. ASCVD is the leading cause of death in both men and women globally, and the population-adjusted risk of ASCVD mortality in women is significantly greater than that in men. Nevertheless, the current focus on the 10-year ASCVD risk estimated by a risk-scoring algorithm such as the Pooled Cohort Equation has shown unsatisfactory accuracy in women. Therefore, new strategies beyond the conventional risk stratification algorithm are needed to improve identification for women at high risk for ASCVD.
ASCVD and osteoporosis are major age-related diseases contributing to significant morbidity and mortality in women, and previous epidemiologic studies have suggested a potential association between these diseases. Given that millions of women are screened for osteoporosis using dual-energy X-ray absorptiometry (DXA), potential associations between low bone mineral density (BMD) and ASCVD in women would provide an opportunity to improve the risk stratification of women without any additional costs. Therefore, we aimed to investigate whether the evaluation of BMD provides independent and incremental prognostic values for ASCVD prediction in women. (more…)