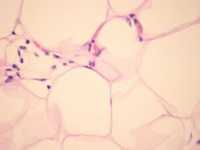
Alcohol, Author Interviews, JAMA, Weight Research / 12.03.2019
Combination of Alcohol and High BMI Linked to Liver Injury Biomarkers
MedicalResearch.com Interview with:
Alice R Carter MSc
Doctor of Philosophy Student
MRC Integrative Epidemiology Unit
Population Health Science, Bristol Medical School
University of Bristol
MedicalResearch.com: What is the background for this study?
Response: Higher body mass index and alcohol intake have been shown to increase the risk of liver disease. Some studies have looked at their combined effect by comparing the risk of liver disease between individuals with both high BMI and high alcohol intake and individuals with low BMI and low alcohol intake. However, these studies have produced mixed results. Some possible reasons for that are errors in self-reported BMI and alcohol intake, other factors confounding the association of BMI & alcohol intake with liver disease risk and changes in lifestyle that individuals with ill health may have been advised to adopt.
One way to overcome these limitations is to use a technique called Mendelian randomisation. This method uses genetic differences between individuals that influence their characteristics (e.g. their body mass and how much alcohol they drink) to help understand whether these characteristics are causally related to diseases.
Our study used this method to explore the joint effects of BMI and alcohol consumption on liver disease and biomarkers of liver injury.



 Dr. Gyamfi-Bannerman[/caption]
Cynthia Gyamfi-Bannerman, MD, MSc
Ellen Jacobson Levine and Eugene Jacobson
Professor of Women's Health in Obstetrics and Gynecology
Director, Maternal-Fetal Medicine Fellowship Program
Co-Director, CUMC Preterm Birth Prevention Center
Columbia University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2016 our group published the findings of the Antenatal Late Preterm Steroids (ALPS) trial in the NEJM. We found that administration of antenatal corticosteroids to women at high risk for delivery from 34-36 weeks decreased breathing problems in their neonates. This treatment had been traditionally only given at less than 34 weeks.
The current paper is a cost analysis of that trial. We found that the treatment was also cost effective. From a cost perspective treatment was both low cost and highly effective (the options are low cost, low effect/low cost/high effect, high cost/low effect, high cost/high effect).
Dr. Gyamfi-Bannerman[/caption]
Cynthia Gyamfi-Bannerman, MD, MSc
Ellen Jacobson Levine and Eugene Jacobson
Professor of Women's Health in Obstetrics and Gynecology
Director, Maternal-Fetal Medicine Fellowship Program
Co-Director, CUMC Preterm Birth Prevention Center
Columbia University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2016 our group published the findings of the Antenatal Late Preterm Steroids (ALPS) trial in the NEJM. We found that administration of antenatal corticosteroids to women at high risk for delivery from 34-36 weeks decreased breathing problems in their neonates. This treatment had been traditionally only given at less than 34 weeks.
The current paper is a cost analysis of that trial. We found that the treatment was also cost effective. From a cost perspective treatment was both low cost and highly effective (the options are low cost, low effect/low cost/high effect, high cost/low effect, high cost/high effect). 

 Dr. Rhee[/caption]
Chanu Rhee, MD,MPH
Assistant Professor of Population Medicine
Harvard Medical School / Harvard Pilgrim Health Care Institute
Assistant Hospital Epidemiologist
Brigham and Women’s Hospital
MedicalResearch.com: What is the background for this study?
Response: Sepsis is the body’s reaction to a serious infection that results a cascade of inflammation in the body and organ dysfunction, such as low blood pressure, confusion, or failure of the lungs, kidneys, or liver. Sepsis is a major cause of death, disability, and cost in the U.S. and around the world. Growing recognition of this problem has led to numerous sepsis performance improvement initiatives in hospitals around the country. Some of these efforts have also been catalyzed by high-profile tragic cases of missed sepsis leading to death, which may have contributed to a perception that most sepsis deaths are preventable if doctors and hospitals were only better at recognizing it.
However, the extent to which sepsis-related deaths might be preventable with better hospital-based care is unknown. In my own experience as a critical care physician, a lot of sepsis patients we treat are extremely sick and even when they receive timely and optimal medical care, many do not survive. This led myself and my colleagues to conduct this study to better understand what types of patients are dying from sepsis and how preventable these deaths might be.
Dr. Rhee[/caption]
Chanu Rhee, MD,MPH
Assistant Professor of Population Medicine
Harvard Medical School / Harvard Pilgrim Health Care Institute
Assistant Hospital Epidemiologist
Brigham and Women’s Hospital
MedicalResearch.com: What is the background for this study?
Response: Sepsis is the body’s reaction to a serious infection that results a cascade of inflammation in the body and organ dysfunction, such as low blood pressure, confusion, or failure of the lungs, kidneys, or liver. Sepsis is a major cause of death, disability, and cost in the U.S. and around the world. Growing recognition of this problem has led to numerous sepsis performance improvement initiatives in hospitals around the country. Some of these efforts have also been catalyzed by high-profile tragic cases of missed sepsis leading to death, which may have contributed to a perception that most sepsis deaths are preventable if doctors and hospitals were only better at recognizing it.
However, the extent to which sepsis-related deaths might be preventable with better hospital-based care is unknown. In my own experience as a critical care physician, a lot of sepsis patients we treat are extremely sick and even when they receive timely and optimal medical care, many do not survive. This led myself and my colleagues to conduct this study to better understand what types of patients are dying from sepsis and how preventable these deaths might be. 
 Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
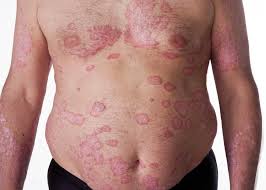
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
 Dr. McCrory[/caption]
Megan A McCrory, PhD, FTOS
Research Associate Professor
Dept of Health Sciences
Sargent College of Health and Rehabilitation Sciences
Boston University 02215
MedicalResearch.com: What is the background for this study?
Response: The prevalence of overweight and obesity has increased in the US, along with documented increases in portion size in the food supply. Fast food is popular, making up about 11% of adult daily calorie intake in the US, and over 1/3 of U.S. adults eat at fast food establishments on any given day. We therefore sought to examine changes in portion size, calories, and selected nutrients in fast-food entree, side, and dessert menu items across the years 1986, 1991, and 2016.
Dr. McCrory[/caption]
Megan A McCrory, PhD, FTOS
Research Associate Professor
Dept of Health Sciences
Sargent College of Health and Rehabilitation Sciences
Boston University 02215
MedicalResearch.com: What is the background for this study?
Response: The prevalence of overweight and obesity has increased in the US, along with documented increases in portion size in the food supply. Fast food is popular, making up about 11% of adult daily calorie intake in the US, and over 1/3 of U.S. adults eat at fast food establishments on any given day. We therefore sought to examine changes in portion size, calories, and selected nutrients in fast-food entree, side, and dessert menu items across the years 1986, 1991, and 2016.





 Dr. Mikkola[/caption]
Tomi Mikkola MD
Associate Professor
Helsinki University Hospital
Department of Obstetrics and Gynecology
Helsinki, Finland
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In Finland we have perhaps the most comprehensive and reliable medical registers in the world. Thus, with my research group I have conducted various large studies evaluating association of postmenopausal hormone therapy use and various major diseases (see e.g. the references in the B;MJ paper). There has been various smaller studies indicating that hormone therapy might be protective for all kinds of dementias, also Alzheimer’s disease.
However, we have quite recently shown that hormone therapy seems to lower the mortality risk of vascular dementia but not Alzheimer’s disease (Mikkola TS et al. J Clin Endocrinol Metab 2017;102:870-7). Now in this upcoming BMJ-paper we report in a very large case-control study (83 688 women with Alzheimer’s disease and same number of control women without the disease) that systemic hormone therapy was associated with a 9-17% increased risk of Alzheimer’s disease.
Furthermore, this risk increase is particularly in women using hormone therapy long, for more than 10 years. This was somewhat surprising finding, but it underlines the fact that mechanisms behind Alzheimer’s disease are likely quite different than in vascular dementia, where the risk factors are similar as in cardiovascular disease. We have also shown how hormone therapy protects against cardiovascular disease, particularly in women who initiate hormone therapy soon after menopause.
Dr. Mikkola[/caption]
Tomi Mikkola MD
Associate Professor
Helsinki University Hospital
Department of Obstetrics and Gynecology
Helsinki, Finland
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In Finland we have perhaps the most comprehensive and reliable medical registers in the world. Thus, with my research group I have conducted various large studies evaluating association of postmenopausal hormone therapy use and various major diseases (see e.g. the references in the B;MJ paper). There has been various smaller studies indicating that hormone therapy might be protective for all kinds of dementias, also Alzheimer’s disease.
However, we have quite recently shown that hormone therapy seems to lower the mortality risk of vascular dementia but not Alzheimer’s disease (Mikkola TS et al. J Clin Endocrinol Metab 2017;102:870-7). Now in this upcoming BMJ-paper we report in a very large case-control study (83 688 women with Alzheimer’s disease and same number of control women without the disease) that systemic hormone therapy was associated with a 9-17% increased risk of Alzheimer’s disease.
Furthermore, this risk increase is particularly in women using hormone therapy long, for more than 10 years. This was somewhat surprising finding, but it underlines the fact that mechanisms behind Alzheimer’s disease are likely quite different than in vascular dementia, where the risk factors are similar as in cardiovascular disease. We have also shown how hormone therapy protects against cardiovascular disease, particularly in women who initiate hormone therapy soon after menopause.




 Dr. Del Rosso[/caption]
Dr. James Q. Del Rosso, D.O., FAOCD, Dermatologist
Research Director and Principal Investigator
Del Rosso Dermatology Research Center, Las Vegas, NV
Galderma Consultant
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Del Rosso[/caption]
Dr. James Q. Del Rosso, D.O., FAOCD, Dermatologist
Research Director and Principal Investigator
Del Rosso Dermatology Research Center, Las Vegas, NV
Galderma Consultant
MedicalResearch.com: What is the background for this study? What are the main findings?