Author Interviews, Gastrointestinal Disease, Immunotherapy / 17.05.2023
Ulcerative Colitis: Lilly Reports Clinically Meaningful Response with Mirikizumab in Phase Two Study
MedicalResearch.com Interview with:
Marla Dubinsky, M.D.
Professor of Pediatrics and Medicine
Co-director of the Susan and Leonard Feinstein IBD Clinical Center
Chief of the Division of Pediatric Gastroenterology and Nutrition
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study? How does MIRIKIZUMAB differ from other medications for UC?
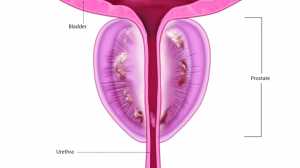
Response: This is a phase 2 study to assess the PK (pharamcokinetics), safety and efficacy of mirikizumab in pediatric ulcerative colitis (UC).
Mirikizumab is a humanized monoclonal antibody that specifically binds to the p19 subunit of interleukin-23, a key inflammatory mediator in inflammatory bowel disease.
(more…)




















 Response: We were broadly interested in discovering instances of bacterial genes that have been acquired by diverse animal genomes over millions of years of evolution by the process of horizontal gene transfer (HGT). Since these events are quite rare and most previous discoveries have been serendipitous, we developed computational methods to identify genes acquired by HGT in animals. One of the exciting discoveries from our work was that vertebrate IRBP appeared to have originated in bacteria and is now a critical component of the vertebrate visual cycle, so this paper focuses on that one discovery.
IRBP or interphotoreceptor retinoid binding protein is an important protein present in the space between two major cell types in our eyes, photoreceptor cells and RPE cells. Our ability to see involves an intricate set of steps where light is first sensed by causing a change (isomerization) in the chemical structure of molecules in the eye called retinoids. This sensing of light occurs in our photoreceptor cells. Following this change in the chemical structure, the retinoid needs to be recycled back to the chemical structure that can again sense light. This recycling occurs in RPE cells. IRBP performs the essential function of shuttling retinoids between the photoreceptors and the RPE cells, which allows the cycle of sensing and regeneration to work. Supporting its importance, mutations in IRBP (also known as retinol binding protein 3 or RBP3) can cause several severe human eye diseases.
Response: We were broadly interested in discovering instances of bacterial genes that have been acquired by diverse animal genomes over millions of years of evolution by the process of horizontal gene transfer (HGT). Since these events are quite rare and most previous discoveries have been serendipitous, we developed computational methods to identify genes acquired by HGT in animals. One of the exciting discoveries from our work was that vertebrate IRBP appeared to have originated in bacteria and is now a critical component of the vertebrate visual cycle, so this paper focuses on that one discovery.
IRBP or interphotoreceptor retinoid binding protein is an important protein present in the space between two major cell types in our eyes, photoreceptor cells and RPE cells. Our ability to see involves an intricate set of steps where light is first sensed by causing a change (isomerization) in the chemical structure of molecules in the eye called retinoids. This sensing of light occurs in our photoreceptor cells. Following this change in the chemical structure, the retinoid needs to be recycled back to the chemical structure that can again sense light. This recycling occurs in RPE cells. IRBP performs the essential function of shuttling retinoids between the photoreceptors and the RPE cells, which allows the cycle of sensing and regeneration to work. Supporting its importance, mutations in IRBP (also known as retinol binding protein 3 or RBP3) can cause several severe human eye diseases.

 Larissa K. Samuelson, PhD
Professor
Developmental Dynamics Lab
School of Psychology; UK 14th for Research Quality
Psychology, Psychiatry, and Neuroscience
University of East Anglia, United Kingdom
MedicalResearch.com: What is the background for this study?
Response: Words direct the attention of infants, children and adults to mentioned objects in the environment. When someone says “Can you find the candy,” you look to the candy sitting on the counter. This fact is the basis of many tests of infant cognition in laboratories. To find out if a child knows the word “bike” we put a picture of a bike and a truck on a TV screen, say the word “bike” and see if they look at the correct object.
There is also evidence that words can direct attention even if you don’t know what they mean yet. For example, in studies of learning in the lab novel made up words like “modi” can direct children’s attention to specific features of objects. One particular example of this is the “shape bias”. If a two-year-old is shown a novel object and told a novel name, for example “This is my blicket,” and then asked, “Can you get your blicket” and shown one object that matches the named one in shape and another that is made from the same material, they will attend to the one that matches in shape. Researchers think the naming event “This is my…” cues children to look at things that are the same shape because they already know many names for things in sets that are similar in shape; cups are all cup-shaped, keys are all key-shaped, spoons are all spoon-shaped, etc.
Prior research suggests there may be differences in the way children who struggle with language decide what a new word means. For example, children with Developmental Language Disorder do not pay attention to the same things when learning new words as children with typical language development. These children do not look to an object that matches a named exemplar in shape when asked to “get your blicket”. But you can’t diagnose children with DLD until they are 3 or 4. We want to see if we can identify these children earlier, so they can get early support.
Larissa K. Samuelson, PhD
Professor
Developmental Dynamics Lab
School of Psychology; UK 14th for Research Quality
Psychology, Psychiatry, and Neuroscience
University of East Anglia, United Kingdom
MedicalResearch.com: What is the background for this study?
Response: Words direct the attention of infants, children and adults to mentioned objects in the environment. When someone says “Can you find the candy,” you look to the candy sitting on the counter. This fact is the basis of many tests of infant cognition in laboratories. To find out if a child knows the word “bike” we put a picture of a bike and a truck on a TV screen, say the word “bike” and see if they look at the correct object.
There is also evidence that words can direct attention even if you don’t know what they mean yet. For example, in studies of learning in the lab novel made up words like “modi” can direct children’s attention to specific features of objects. One particular example of this is the “shape bias”. If a two-year-old is shown a novel object and told a novel name, for example “This is my blicket,” and then asked, “Can you get your blicket” and shown one object that matches the named one in shape and another that is made from the same material, they will attend to the one that matches in shape. Researchers think the naming event “This is my…” cues children to look at things that are the same shape because they already know many names for things in sets that are similar in shape; cups are all cup-shaped, keys are all key-shaped, spoons are all spoon-shaped, etc.
Prior research suggests there may be differences in the way children who struggle with language decide what a new word means. For example, children with Developmental Language Disorder do not pay attention to the same things when learning new words as children with typical language development. These children do not look to an object that matches a named exemplar in shape when asked to “get your blicket”. But you can’t diagnose children with DLD until they are 3 or 4. We want to see if we can identify these children earlier, so they can get early support.