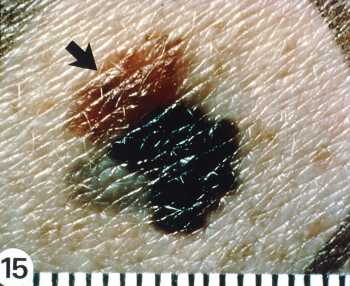
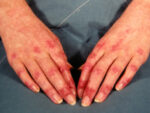
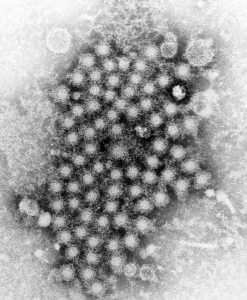
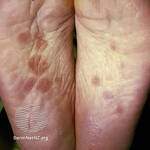
Author Interviews, Dermatology / 22.11.2022
Makeup Applied to Facial Features Improves Perceived Skin Evenness
MedicalResearch.com Interview with:
Carlota Batres, Ph.D.Assistant Professor, Department of PsychologyDirector, Preferences Lab
PreferencesLab.comFranklin and Marshall College
MedicalResearch.com: What is the background for this study?
Response: Previous research has found that complexion-oriented makeup products, such as foundation and concealer, make the skin appear more even. Interestingly, though, the effect size of perceptual judgements has been found to be larger than the effect size of physical measurements, suggesting that there are factors affecting the perception of skin evenness that are not captured by the physical measurements of isolated skin patches.
(more…)




 Response: We started many years ago by studying some natural and synthetic molecules that were able to counteract the early stages of this disease. With a view to early prevention, we wondered if these molecules could be routinely taken with the diet or dietary supplements. So our studies focused on the search for bioactive molecules present in food and edible plants.
Hops are very rich in polyphenolic compounds, and for this reason it is historically used for the production of beer, but also for the preparation of herbal teas and infusions, and its uses in traditional medicine are known. In recent years we have also collected interesting results on Coffee, Sage, Radix Imperatoriae, Cocoa and Cinnamon extracts.
Response: We started many years ago by studying some natural and synthetic molecules that were able to counteract the early stages of this disease. With a view to early prevention, we wondered if these molecules could be routinely taken with the diet or dietary supplements. So our studies focused on the search for bioactive molecules present in food and edible plants.
Hops are very rich in polyphenolic compounds, and for this reason it is historically used for the production of beer, but also for the preparation of herbal teas and infusions, and its uses in traditional medicine are known. In recent years we have also collected interesting results on Coffee, Sage, Radix Imperatoriae, Cocoa and Cinnamon extracts.







 Sean M Hughes MA
Department of Obstetrics and Gynecology
University of Washington
Seattle, WA
MedicalResearch.com: What is the background for this study?
Response: Young women are at elevated risk of getting sexually transmitted infections at the age when they typically start to have sexual intercourse. It’s not known whether this elevated risk is a consequence of behavioral factors (such as choices around use of barrier protection), physiological factors (such as a difference in the immune system) or a combination of both. In this study, we investigated a physiological factor: the immune system in the vagina.
Sean M Hughes MA
Department of Obstetrics and Gynecology
University of Washington
Seattle, WA
MedicalResearch.com: What is the background for this study?
Response: Young women are at elevated risk of getting sexually transmitted infections at the age when they typically start to have sexual intercourse. It’s not known whether this elevated risk is a consequence of behavioral factors (such as choices around use of barrier protection), physiological factors (such as a difference in the immune system) or a combination of both. In this study, we investigated a physiological factor: the immune system in the vagina.