Author Interviews, Cancer Research, FDA, Vaccine Studies / 14.08.2019
FDA Identifies No New Safety Concerns Associated with Prostate Cancer Treatment Vaccine Sipuleucel-T
MedicalResearch.com Interview with:
[caption id="attachment_50871" align="alignleft" width="150"] Dr. Graca Dores (left) and Dr. Perez-Vilar (senior author)[/caption]
Dr. Graca Dores MD MPH
US Food and Drug Administration
Center for Biologics Evaluation and Research
Office of Biostatistics and Epidemiology
Division of Epidemiology
Silver Spring, Maryland
Oklahoma City, OK
MedicalResearch.com: What is the background for this study? Would you briefly explain what Sipuleucel-T is used for?
Response: Sipuleucel-T was the first therapeutic vaccine approved by the U.S. Food and Drug Administration (FDA) in 2010. It is indicated for the treatment of asymptomatic or minimally symptomatic, metastatic, castration-resistant prostate cancer (CRPC; prostate cancer that spreads while an individual is on hormone-blocking therapy). During the preparation of this product, the patient’s cells are collected (leukapheresis), sent for processing to generate a dose of patient-specific vaccine, and then administered intravenously back to the patient. This process is repeated approximately every two weeks for a total of three doses.
Except for the pre-marketing clinical trials that were reviewed during the sipuleucel-T approval process, post-marketing studies that have evaluated the safety profile of sipuleucel-T are scarce. Therefore, we used the FDA’s Adverse Event Reporting System (FAERS) database to summarize the adverse events reported to FDA by industry, medical professionals, and consumers. We also assessed whether sipuleucel-T and specific adverse events (product-event pairs) were reported more than expected compared to all other drug/biologic-adverse event pairs in the FAERS database.
Dr. Graca Dores (left) and Dr. Perez-Vilar (senior author)[/caption]
Dr. Graca Dores MD MPH
US Food and Drug Administration
Center for Biologics Evaluation and Research
Office of Biostatistics and Epidemiology
Division of Epidemiology
Silver Spring, Maryland
Oklahoma City, OK
MedicalResearch.com: What is the background for this study? Would you briefly explain what Sipuleucel-T is used for?
Response: Sipuleucel-T was the first therapeutic vaccine approved by the U.S. Food and Drug Administration (FDA) in 2010. It is indicated for the treatment of asymptomatic or minimally symptomatic, metastatic, castration-resistant prostate cancer (CRPC; prostate cancer that spreads while an individual is on hormone-blocking therapy). During the preparation of this product, the patient’s cells are collected (leukapheresis), sent for processing to generate a dose of patient-specific vaccine, and then administered intravenously back to the patient. This process is repeated approximately every two weeks for a total of three doses.
Except for the pre-marketing clinical trials that were reviewed during the sipuleucel-T approval process, post-marketing studies that have evaluated the safety profile of sipuleucel-T are scarce. Therefore, we used the FDA’s Adverse Event Reporting System (FAERS) database to summarize the adverse events reported to FDA by industry, medical professionals, and consumers. We also assessed whether sipuleucel-T and specific adverse events (product-event pairs) were reported more than expected compared to all other drug/biologic-adverse event pairs in the FAERS database.
 Dr. Graca Dores (left) and Dr. Perez-Vilar (senior author)[/caption]
Dr. Graca Dores MD MPH
US Food and Drug Administration
Center for Biologics Evaluation and Research
Office of Biostatistics and Epidemiology
Division of Epidemiology
Silver Spring, Maryland
Oklahoma City, OK
MedicalResearch.com: What is the background for this study? Would you briefly explain what Sipuleucel-T is used for?
Response: Sipuleucel-T was the first therapeutic vaccine approved by the U.S. Food and Drug Administration (FDA) in 2010. It is indicated for the treatment of asymptomatic or minimally symptomatic, metastatic, castration-resistant prostate cancer (CRPC; prostate cancer that spreads while an individual is on hormone-blocking therapy). During the preparation of this product, the patient’s cells are collected (leukapheresis), sent for processing to generate a dose of patient-specific vaccine, and then administered intravenously back to the patient. This process is repeated approximately every two weeks for a total of three doses.
Except for the pre-marketing clinical trials that were reviewed during the sipuleucel-T approval process, post-marketing studies that have evaluated the safety profile of sipuleucel-T are scarce. Therefore, we used the FDA’s Adverse Event Reporting System (FAERS) database to summarize the adverse events reported to FDA by industry, medical professionals, and consumers. We also assessed whether sipuleucel-T and specific adverse events (product-event pairs) were reported more than expected compared to all other drug/biologic-adverse event pairs in the FAERS database.
Dr. Graca Dores (left) and Dr. Perez-Vilar (senior author)[/caption]
Dr. Graca Dores MD MPH
US Food and Drug Administration
Center for Biologics Evaluation and Research
Office of Biostatistics and Epidemiology
Division of Epidemiology
Silver Spring, Maryland
Oklahoma City, OK
MedicalResearch.com: What is the background for this study? Would you briefly explain what Sipuleucel-T is used for?
Response: Sipuleucel-T was the first therapeutic vaccine approved by the U.S. Food and Drug Administration (FDA) in 2010. It is indicated for the treatment of asymptomatic or minimally symptomatic, metastatic, castration-resistant prostate cancer (CRPC; prostate cancer that spreads while an individual is on hormone-blocking therapy). During the preparation of this product, the patient’s cells are collected (leukapheresis), sent for processing to generate a dose of patient-specific vaccine, and then administered intravenously back to the patient. This process is repeated approximately every two weeks for a total of three doses.
Except for the pre-marketing clinical trials that were reviewed during the sipuleucel-T approval process, post-marketing studies that have evaluated the safety profile of sipuleucel-T are scarce. Therefore, we used the FDA’s Adverse Event Reporting System (FAERS) database to summarize the adverse events reported to FDA by industry, medical professionals, and consumers. We also assessed whether sipuleucel-T and specific adverse events (product-event pairs) were reported more than expected compared to all other drug/biologic-adverse event pairs in the FAERS database.











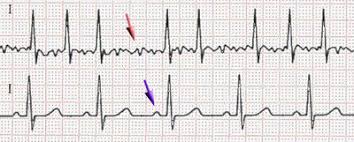







 Dr. Phelan[/caption]
Dermot Phelan, MD, PhD
Director of the Sports Cardiology Center
Cleveland Clinic in Cleveland, Ohio
MedicalResearch.com: What is the background for this study?
Response: It is well recognized that long-term elite endurance athletes are at higher risk of atrial fibrillation. We wished to evaluate whether this held true for primarily strength-type athletes. We had the opportunity to screen almost 500 former NFL athletes. It became clear that we were seeing more atrial fibrillation than one would expect during the screenings.
Dr. Phelan[/caption]
Dermot Phelan, MD, PhD
Director of the Sports Cardiology Center
Cleveland Clinic in Cleveland, Ohio
MedicalResearch.com: What is the background for this study?
Response: It is well recognized that long-term elite endurance athletes are at higher risk of atrial fibrillation. We wished to evaluate whether this held true for primarily strength-type athletes. We had the opportunity to screen almost 500 former NFL athletes. It became clear that we were seeing more atrial fibrillation than one would expect during the screenings.







 Dr. Wee[/caption]
Christina C. Wee, MD, MPH
Associate Professor of Medicine
Harvard Medical School
Director , Obesity Research Program Division of General Medicine
Beth Israel Deaconess Medical Center (BIDMC)
Associate Program Director, Internal Medicine Program, BIDMC
Deputy Editor of the Annals of Internal Medicine
MedicalResearch.com: What is the background for this study?
Response: New research is showing that for many people without diagnosed heart disease, the risk of bleeding may outweigh the benefits of taking a daily aspirin particularly in adults over 70 years of age. The American Heart Association and the American College of Cardiology recently updated their guidelines and now explicitly recommend against aspirin use among those over the age of 70 who do not have existing heart disease or stroke.
Our study found that in 2017, a quarter of adults aged 40 years or older without cardiovascular disease – approximately 29 million people – reported taking daily aspirin for prevention of heart disease. Of these, some 6.6. million people did so without a physician's recommendation.
Dr. Wee[/caption]
Christina C. Wee, MD, MPH
Associate Professor of Medicine
Harvard Medical School
Director , Obesity Research Program Division of General Medicine
Beth Israel Deaconess Medical Center (BIDMC)
Associate Program Director, Internal Medicine Program, BIDMC
Deputy Editor of the Annals of Internal Medicine
MedicalResearch.com: What is the background for this study?
Response: New research is showing that for many people without diagnosed heart disease, the risk of bleeding may outweigh the benefits of taking a daily aspirin particularly in adults over 70 years of age. The American Heart Association and the American College of Cardiology recently updated their guidelines and now explicitly recommend against aspirin use among those over the age of 70 who do not have existing heart disease or stroke.
Our study found that in 2017, a quarter of adults aged 40 years or older without cardiovascular disease – approximately 29 million people – reported taking daily aspirin for prevention of heart disease. Of these, some 6.6. million people did so without a physician's recommendation.

 Frank Qian[/caption]
Frank Qian, MPH
Department of Nutrition
Harvard T. H. Chan School of Public Health
Boston, Massachusetts
MedicalResearch.com: What is the background for this study?
Response: Plant-based diets have really grown in popularity in the last several years, particularly among the younger generation in the United States, many of whom are adopting a plant-based or vegetarian/vegan diet. However, the quality of such a diet can vary drastically. While many prior studies have demonstrated beneficial associations for risk of type 2 diabetes with healthful plant-based foods such as fruits, vegetables, nuts/seeds, whole grains, and legumes, the opposite is true for less healthful plant-based foods such as potatoes and refined grains such as white rice. In addition, some animal-based foods, such as dairy and fish, have shown protective associations against the development of type 2 diabetes, so strict vegetarian diets which exclude these foods may miss out on the potential benefits.
Given these divergent findings, we sought to pool all the available data from prior cohort studies to analyze whether the overall association of a diet which emphasizes plant-based foods (both healthful and unhealthful) are related to risk of type 2 diabetes.
Frank Qian[/caption]
Frank Qian, MPH
Department of Nutrition
Harvard T. H. Chan School of Public Health
Boston, Massachusetts
MedicalResearch.com: What is the background for this study?
Response: Plant-based diets have really grown in popularity in the last several years, particularly among the younger generation in the United States, many of whom are adopting a plant-based or vegetarian/vegan diet. However, the quality of such a diet can vary drastically. While many prior studies have demonstrated beneficial associations for risk of type 2 diabetes with healthful plant-based foods such as fruits, vegetables, nuts/seeds, whole grains, and legumes, the opposite is true for less healthful plant-based foods such as potatoes and refined grains such as white rice. In addition, some animal-based foods, such as dairy and fish, have shown protective associations against the development of type 2 diabetes, so strict vegetarian diets which exclude these foods may miss out on the potential benefits.
Given these divergent findings, we sought to pool all the available data from prior cohort studies to analyze whether the overall association of a diet which emphasizes plant-based foods (both healthful and unhealthful) are related to risk of type 2 diabetes.








